Nomenclature of coordination compounds - Free MCQ Practice Test with solutions,
MCQ Practice Test & Solutions: Test: Nomenclature of coordination compounds (20 Questions)
You can prepare effectively for JEE Chemistry for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Nomenclature of coordination compounds". These 20 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 20 minutes
- - Number of Questions: 20
Sign up on EduRev for free to attempt this test and track your preparation progress.
Detailed Solution: Question 1
select the correct IUPAC name for the following:
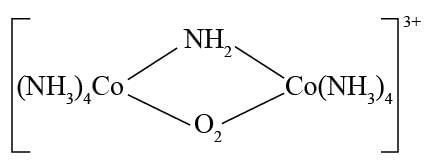
Detailed Solution: Question 2
Detailed Solution: Question 3
The correct IUPAC name of the complex:
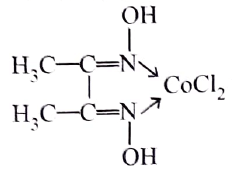
is
Detailed Solution: Question 4
Coordination number and oxidation state of 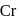 in
in 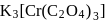 are, respectively:
are, respectively:
Detailed Solution: Question 5
Detailed Solution: Question 6
Which of the following is not a complex salt 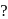
Detailed Solution: Question 7
Detailed Solution: Question 8
Red precipitate is obtained when ethanol solution of dimethylglyoxime is added to ammoniacal Ni(II). Which of the following statements is not true?
Detailed Solution: Question 9
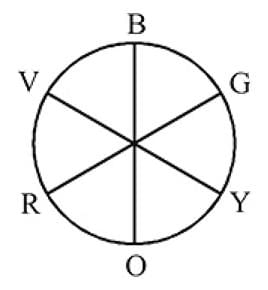
The octahedral complex of a metal ion 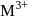 with four monodentate ligands
with four monodentate ligands 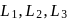 and
and 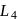 absorb wavelengths in the region of red, green, yellow and blue, respectively. The increasing order of ligand strength of the four ligands is:
absorb wavelengths in the region of red, green, yellow and blue, respectively. The increasing order of ligand strength of the four ligands is:
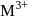 with four monodentate ligands
with four monodentate ligands 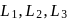 and
and 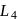 absorb wavelengths in the region of red, green, yellow and blue, respectively. The increasing order of ligand strength of the four ligands is:
absorb wavelengths in the region of red, green, yellow and blue, respectively. The increasing order of ligand strength of the four ligands is:Detailed Solution: Question 10
Which one of the following coordination compounds is used to inhibit the growth of tumours?
Detailed Solution: Question 11
The EAN of iron in potassium ferricyanide is
Detailed Solution: Question 12
AgCl is soluble in 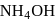 solution. The solubility is due to formation of
solution. The solubility is due to formation of
Detailed Solution: Question 13
In which of the following coordinate compounds the central metal atom obeys the EAN rule.
Detailed Solution: Question 14
Which of the following will exhibit maximum ionic conductivity?
Detailed Solution: Question 15
Which of the following complexes have a maximum number of unpaired electrons?
Detailed Solution: Question 16
The degeneracy of 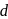 -orbitals is lost under
-orbitals is lost under
(I) Strong field ligand
(II) Weak field ligand
(III) Mixed field ligand
(IV) Chelated ligand field
Detailed Solution: Question 17
The IUPAC name of the following complex is 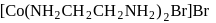
Detailed Solution: Question 18
Which of the following name formula combinations is not correct?
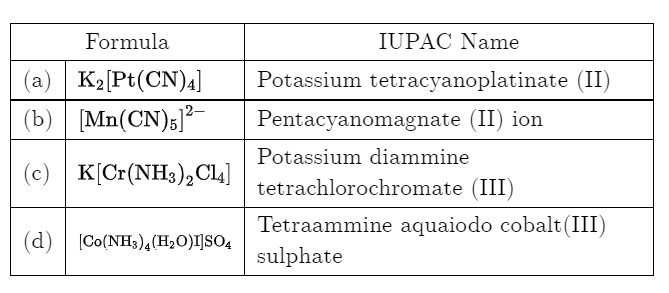
Detailed Solution: Question 19
In 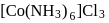 , the number of covalent bonds is
, the number of covalent bonds is
Detailed Solution: Question 20
335 videos|697 docs|300 tests |


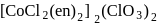 .
. -Amido-
-Amido-  -peroxidobis(tetraammine)dicobalt-(III) ion
-peroxidobis(tetraammine)dicobalt-(III) ion
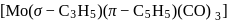
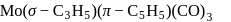 :
: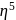 - cyclopentadienyl) molybdenum (II).
- cyclopentadienyl) molybdenum (II).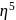 represents that the cyclopnetadienyl ligand shares 5 electrons with metal.
represents that the cyclopnetadienyl ligand shares 5 electrons with metal. is 6 .
is 6 .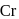 be
be 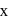 .
.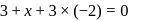
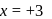
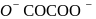 ?
?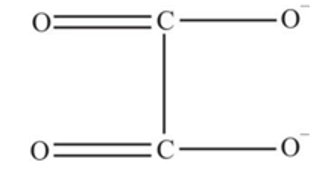
 oxalate ion is bidentate due to two donor atom
oxalate ion is bidentate due to two donor atom 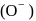
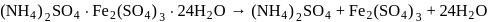
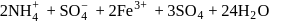
 - bonded complex ?
- bonded complex ?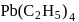 is not
is not 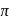 bonded complex. It is
bonded complex. It is 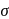 bonded organometallic compound
bonded organometallic compound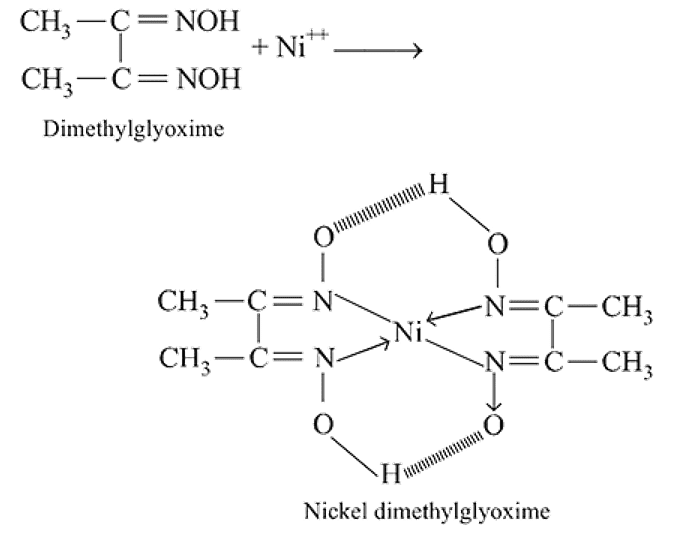
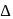 , which will absorbs light of longer
, which will absorbs light of longer 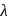 and thus lower frequency. Conservely, stronger field ligands create a larger
and thus lower frequency. Conservely, stronger field ligands create a larger 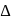 , absorb light of shorter
, absorb light of shorter 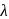 and thus higher
and thus higher 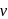 i.e. higher energy.
i.e. higher energy.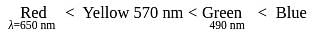
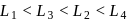
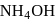 which is
which is 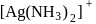
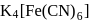 obeys the EAN rule.
obeys the EAN rule.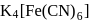 in solution furnishes
in solution furnishes 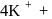
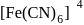 five ions and exhibits maximum ionic conductivity.
five ions and exhibits maximum ionic conductivity.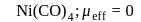
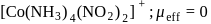
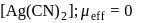
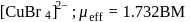
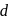 -orbitals undergo splitting under ligand field created by strong, weak or mixed ligands.
-orbitals undergo splitting under ligand field created by strong, weak or mixed ligands.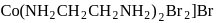 is dibromidobis (ethylene diamine) cobalt (III) bromide.
is dibromidobis (ethylene diamine) cobalt (III) bromide.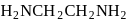 is ethylene diamine which is a bidentate ligand.
is ethylene diamine which is a bidentate ligand.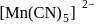 Pentacyanomangnate (III).
Pentacyanomangnate (III). and
and  . Therefore the number of covalent bonds is 18 .
. Therefore the number of covalent bonds is 18 .