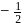Oxidation and Reduction Reactions, number - Free MCQ Practice Test
MCQ Practice Test & Solutions: Test: Oxidation and Reduction Reactions, Oxidation number (10 Questions)
You can prepare effectively for JEE Chemistry for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Oxidation and Reduction Reactions, Oxidation number". These 10 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 20 minutes
- - Number of Questions: 10
Sign up on EduRev for free to attempt this test and track your preparation progress.
Which one of the following cannot function as an oxidising agent?
Detailed Solution: Question 1
Which of the following chemical reactions depict the oxidizing beahviour of 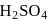 ?
?
Detailed Solution: Question 2
Which reaction involves neither oxidation nor reduction?
Detailed Solution: Question 3
One gas bleaches the colour of flowers by reduction, while the other by oxidation
Detailed Solution: Question 4
In how many of the following species, an element has fractional oxidation state?
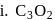
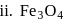
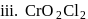
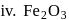
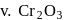
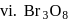
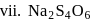
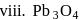
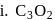
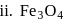
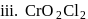
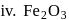
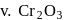
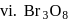
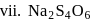
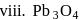
Detailed Solution: Question 5
The most powerful reducing agent is:
Detailed Solution: Question 6
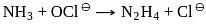 On balancing the above equation in basic solution, using integral coefficient, which of the following whole numbers will be the coefficient of
On balancing the above equation in basic solution, using integral coefficient, which of the following whole numbers will be the coefficient of 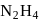 ?
?
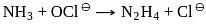 On balancing the above equation in basic solution, using integral coefficient, which of the following whole numbers will be the coefficient of
On balancing the above equation in basic solution, using integral coefficient, which of the following whole numbers will be the coefficient of 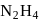 ?
?Detailed Solution: Question 7
In 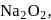 the oxidation states respectively for
the oxidation states respectively for 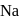 and oxygen may be:
and oxygen may be:
Detailed Solution: Question 8
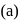
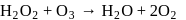
Role of hydrogen peroxide in the above reactions is respectively
Detailed Solution: Question 9
Oxidation number of potassium in 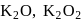 and
and 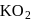 , respectively:
, respectively:
Detailed Solution: Question 10
335 videos|699 docs|300 tests |


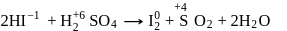
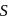 is decreasing from
is decreasing from 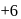 to
to 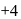 hence undergoing reduction and for HI oxidation Number of I is increasing from
hence undergoing reduction and for HI oxidation Number of I is increasing from 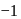 to 0 hence underegoing oxidation therefore
to 0 hence underegoing oxidation therefore 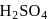 is acting as oxidising agent.
is acting as oxidising agent.
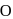 .
. 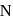 . of
. of 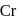 on both sides is
on both sides is 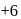 .
.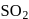 bleaches by reduction, while chlorine bleaches colour of flowers by oxidation.
bleaches by reduction, while chlorine bleaches colour of flowers by oxidation.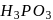 oxidation state of P is+3 , which can further be oxidized to a higher oxidation state. So they can act as reducing agents. In
oxidation state of P is+3 , which can further be oxidized to a higher oxidation state. So they can act as reducing agents. In 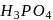 oxidation state of P is +5, which is highest for P. Hence
oxidation state of P is +5, which is highest for P. Hence 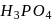 cannot serve as a reducing agent.
cannot serve as a reducing agent.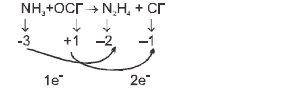
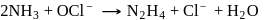
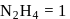
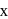 be the oxidation state of
be the oxidation state of 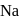 in
in 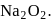
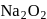 contains a peroxide linkage in which
contains a peroxide linkage in which 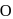 has an
has an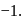
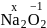 or
or 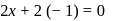 or
or 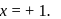
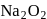 is
is 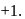
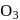 and
and 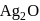 are reduced.
are reduced. 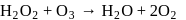
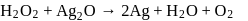
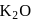 (oxide) is -2, and in
(oxide) is -2, and in 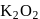 (peroxide) is -1 and in
(peroxide) is -1 and in 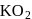 (superoxide) is
(superoxide) is