Oxides & Oxo Anions of Cr Mn - Free MCQ Practice Test with solutions, NEET
MCQ Practice Test & Solutions: Test: Oxides & Oxo Anions of Cr & Mn (25 Questions)
You can prepare effectively for NEET NCERT Based Tests for NEET with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Oxides & Oxo Anions of Cr & Mn". These 25 questions have been designed by the experts with the latest curriculum of NEET 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 35 minutes
- - Number of Questions: 25
Sign up on EduRev for free to attempt this test and track your preparation progress.
Only One Option Correct Type
Direction (Q. Nos. 1-10) This section contains 10 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONLY ONE is correct.
Q.
Which is most basic oxide of chromium?
Detailed Solution: Question 1
The basic character of the transition metal monoxides follows the order
(Atomic Number, Ti = 22, V = 23, Cr = 24, Fe = 26)
Detailed Solution: Question 2
MnO4- + S2- + H+ → Mn2+ +S + H2O When this equation is balanced, the total number of coefficients on the left hand side are
Detailed Solution: Question 3
 when this equation is balanced, the num ber of OH- ions added
when this equation is balanced, the num ber of OH- ions added
Detailed Solution: Question 4
One mole of each KMnO4 and K2Cr2O7 can oxidise ........... moles of ferrous ion.
Detailed Solution: Question 5
 is of intense pink colour, though Mn is in (+7) oxidation state. It is due to
is of intense pink colour, though Mn is in (+7) oxidation state. It is due to
Detailed Solution: Question 6
Which of the following is incorrect statement?
Detailed Solution: Question 7
Pyrolusite in MnO2 is used to prepare KMnO4. Steps are
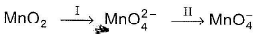
Here, I and II are
Detailed Solution: Question 8
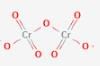
Which is the correct statem ent about 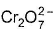 structure?
structure?
Detailed Solution: Question 9
Out of Cr (VI) as 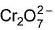
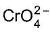 , which is better oxidising agent?
, which is better oxidising agent?
Detailed Solution: Question 10
One or More than One Options Correct Type
Direction (Q. Nos. 11-15) This section contains 5 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONE or MORE THAN ONE are correct.
Q.
Which of the following statem ents is correct when a mixture of NaCI and K2Cr2O7 is gently warmed with cone. H2SO4?
Detailed Solution: Question 11
Detailed Solution: Question 12
Which of the following on heating produce oxygen gas?
Detailed Solution: Question 13
Which of the following statements is not true?
Detailed Solution: Question 14
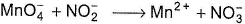 when this redox reaction is balanced in acidic medium, we get
when this redox reaction is balanced in acidic medium, we get
Detailed Solution: Question 15
Comprehension Type
Direction (Q. Nos. 16 and 17) This section contains a paragraph, describing theory, experiments, data, etc. Two questions related to the paragraph have been given. Each question has only one correct answer among the four given options (a), (b), (c) and (d).
Passage
Oxides are generally formed by the reaction of metals with oxygen at high temperatures. All the metals except scandium form MO oxides which are ionic. The highest oxidation number in the oxides, coincides with the group number and is attained in Sc2O3 to Mn2O7. Beyond 7th group, higher oxides coinciding with the group number do not form. As the oxidation number of metal increases covalent character and acidity increases. Mn2O7, CrO3 are acidic and their lower oxides are basic and amphoteric. Stability of higher oxides increases down the group.
Q.
Which of the following reacts with both acids and bases?
Detailed Solution: Question 16
Oxides are generally formed by the reaction of metals with oxygen at high temperatures. All the metals except scandium form MO oxides which are ionic. The highest oxidation number in the oxides, coincides with the group number and is attained in Sc2O3 to Mn2O7. Beyond 7th group, higher oxides coinciding with the group number do not form. As the oxidation number of metal increases covalent character and acidity increases. Mn2O7, CrO3 are acidic and their lower oxides are basic and amphoteric. Stability of higher oxides increases down the group.
Q.
Which of the following undergo disproportionation?
Detailed Solution: Question 17
Matching List Type
Direction (Q. Nos. 18 and 19) Choices for the correct combination of elements from Column I and Column II are given as options (a), (b), (c) and (d), out of which one is correct.
Q.
Match the Column I with Column II and mark the correct option from the codes given below.

Detailed Solution: Question 18
Match the Column I with Column II and mark the correct option from the codes given below.

Detailed Solution: Question 19
One Integer Value Correct Type
Direction (Q. Nos. 20-23) This section contains 4 questions. When worked out will result in an integer from 0 to 9 (both inclusive
Q.
The number of moles of FeC2O4 that reduces two moles of KMnO4 in acidic medium are
Detailed Solution: Question 20
50 cc of 0.04 M K2Cr2O7 in acidic medium oxidises a sample of H2S gas to sulphur. Volume of 0.03 M KMnO4 required to oxidise the same amount of H2S gas to sulphur in acidic medium is 10 x x. Here, the value of x is
Detailed Solution: Question 21
Detailed Solution: Question 22
If molarity of K2Cr2O7 solution is 0.5 M, its normality value is
Detailed Solution: Question 23
Statement Type
Direction (Q. Nos. 24 and 25) This section is based on Statement I and Statement II. Select the correct answer from the codes given below.
Q.
Statement I ; When a solution of potassium chromate is treated with an excess of dilute nitric acid chromate undergoes oxidation.
Statement II : Dichromate ions are produced in above process.
Detailed Solution: Question 24
Statement I : The oxidation state of chromium in the final product formed by the reaction between Kl and acidified potassium dichromate solution has green colour.
Statement II : The product has Cr3+ ions.
Detailed Solution: Question 25