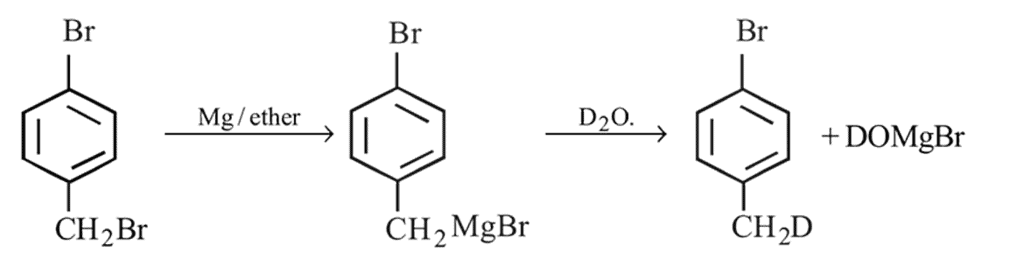
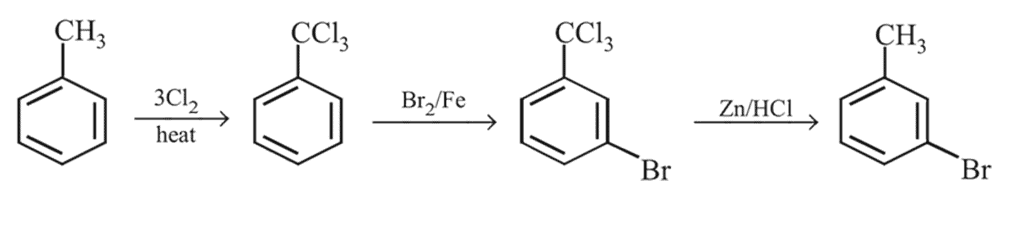
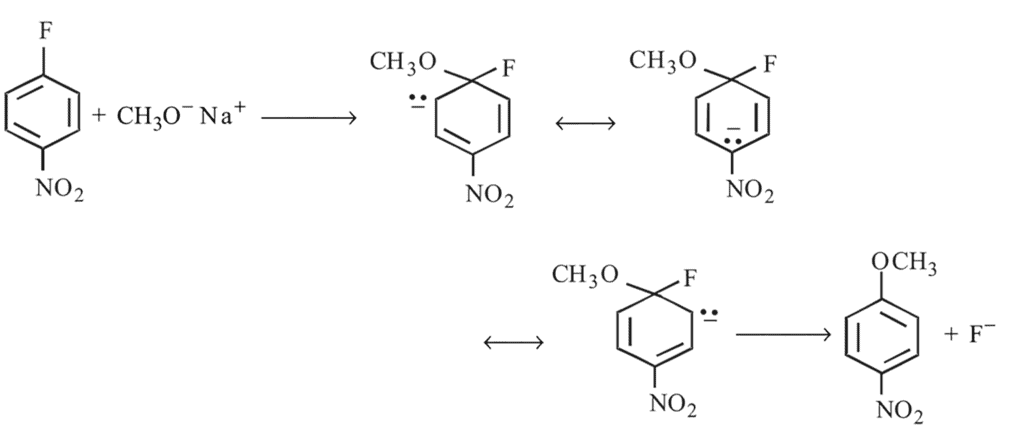
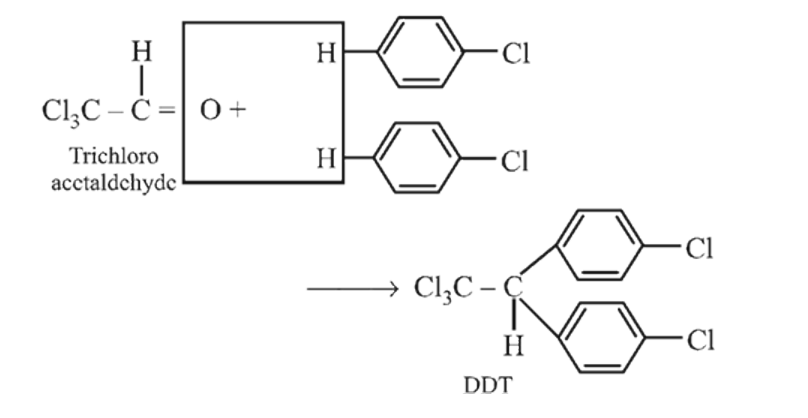
Preparation and Properties of Aryl Halides,Geminal Vicinal Dihalides -
MCQ Practice Test & Solutions: Test: Preparation and Properties of Aryl Halides,Geminal and Vicinal Dihalides (15 Questions)
You can prepare effectively for JEE Chemistry for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Preparation and Properties of Aryl Halides,Geminal and Vicinal Dihalides". These 15 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 20 minutes
- - Number of Questions: 15
Sign up on EduRev for free to attempt this test and track your preparation progress.
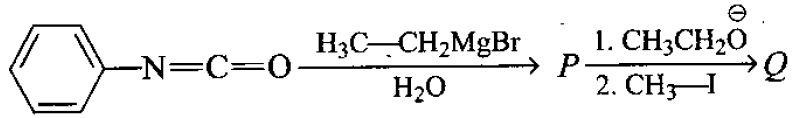
Find out  and
and  :
:
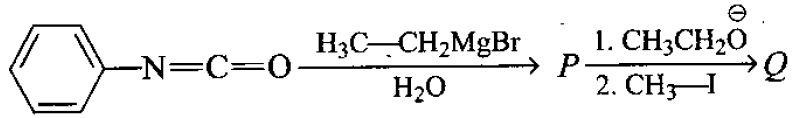
Find out
 and
and  :
:Detailed Solution: Question 1
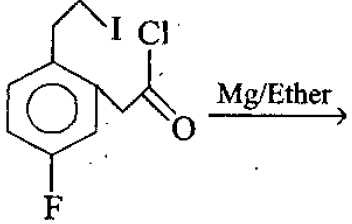
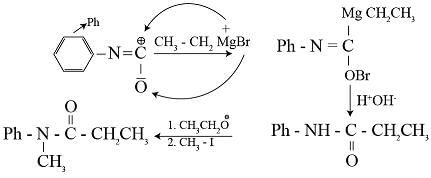
The final product of the reaction is :

The final product of the reaction is :
Detailed Solution: Question 2
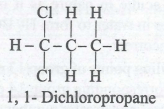
Which of the following is the geminal dichloride?
Detailed Solution: Question 3
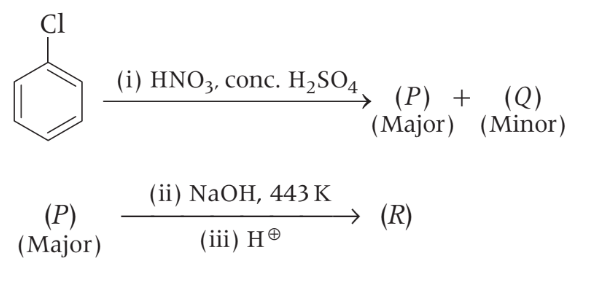
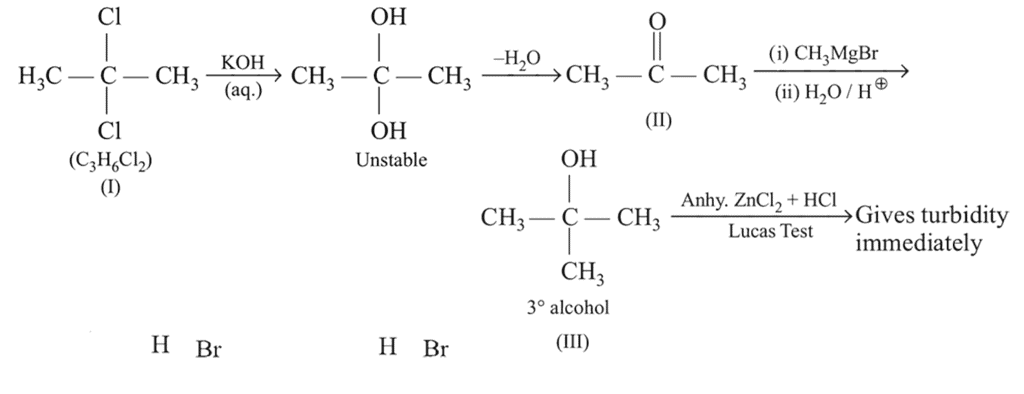
In the following reaction sequence :
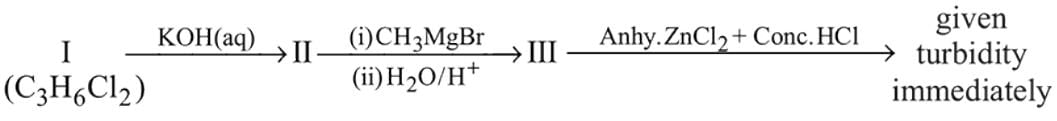
The compound I is :

The compound I is :
Detailed Solution: Question 4
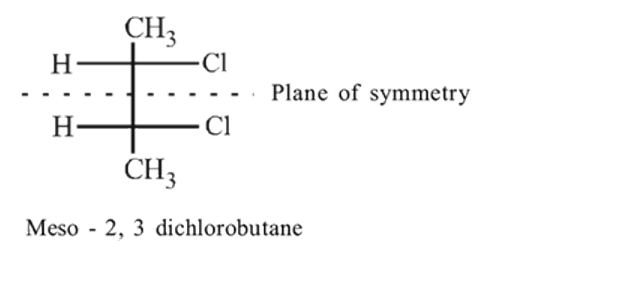
Which of the following will have a meso isomer?
Detailed Solution: Question 5
Identify 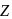 in the reaction :
in the reaction :
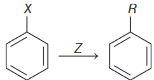
Detailed Solution: Question 6
Arrange the following compounds in order of increasing dipole moment :
(I) Toluene
(II) 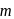 -dichlorobenzene
-dichlorobenzene
(III) 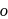 -dichlorobenzene
-dichlorobenzene
(IV) 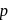 -dichlorobenzene
-dichlorobenzene
Detailed Solution: Question 7
The product 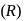 in the following synthetic scheme is
in the following synthetic scheme is
Detailed Solution: Question 8
Which of the following statements are correct?
A. The 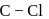 bond in chlorobenzene is shorter than in chloromethane.
bond in chlorobenzene is shorter than in chloromethane.
B. It is difficult to replace chlorine from chlorobenzene than from benzyl chloride.
C. The 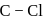 bond in chlorobenzene has some double bond character.
bond in chlorobenzene has some double bond character.
D. Chlorobenzene on chlorination gives m-dichlorobenzene
Detailed Solution: Question 9
Identify the halogen exchange reaction from the following.
Detailed Solution: Question 10
Replacement of 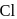 of chlorobenzene to give phenol requires drastic conditions but chlorine of 2,4 -dinitrochlorobenzene is readily replaced This is because
of chlorobenzene to give phenol requires drastic conditions but chlorine of 2,4 -dinitrochlorobenzene is readily replaced This is because
Detailed Solution: Question 11
The final product obtained in the reaction
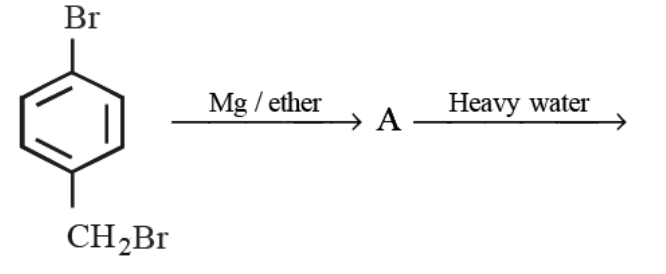
Detailed Solution: Question 12
The compound
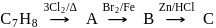
The compound 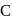 is
is
Detailed Solution: Question 13
Which of the following is (are) true concerning the intermediate in the addition-elimination mechanism of the following reaction ?
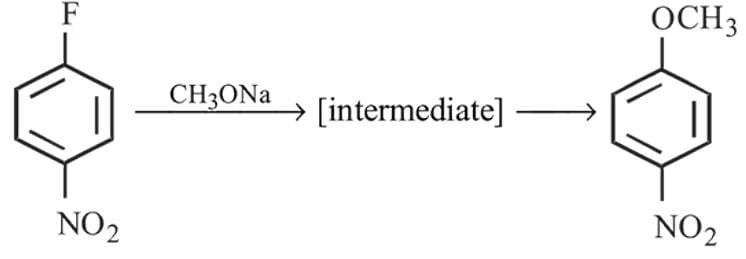
 The intermediate is aromatic,
The intermediate is aromatic,  The intermediate is a resonance stabilised anion,
The intermediate is a resonance stabilised anion, 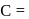 Electron withdrawing groups on the benzene ring stabilises the intermediate
Electron withdrawing groups on the benzene ring stabilises the intermediate

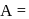 The intermediate is aromatic,
The intermediate is aromatic, 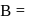 The intermediate is a resonance stabilised anion,
The intermediate is a resonance stabilised anion, 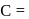 Electron withdrawing groups on the benzene ring stabilises the intermediate
Electron withdrawing groups on the benzene ring stabilises the intermediateDetailed Solution: Question 14
Chlorobenzene reacts with trichloro acetaldehyde in the presence of 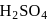 .
.
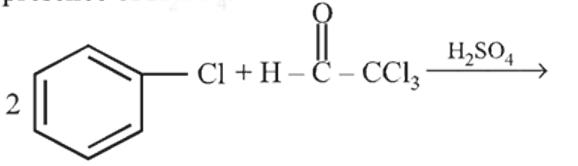
The major product formed is:
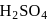 .
.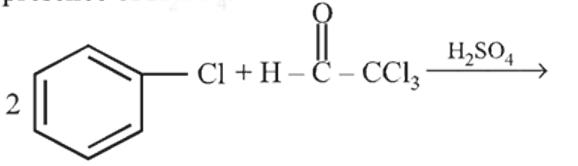
The major product formed is:
Detailed Solution: Question 15
335 videos|699 docs|300 tests |



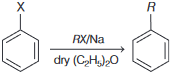
 -dichlorobenzene, the two equal dipoles are in opposite direction, hence the molecule has zero dipole moment. In
-dichlorobenzene, the two equal dipoles are in opposite direction, hence the molecule has zero dipole moment. In  - and
- and  - dichlorobenzenes, the two dipoles are at
- dichlorobenzenes, the two dipoles are at 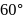 and
and 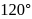 apart respectively, and thus according to parallelogram law of forces, the dipole moment of
apart respectively, and thus according to parallelogram law of forces, the dipole moment of 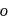 -dichlorobenzene is much higher than that of
-dichlorobenzene is much higher than that of 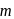 -isomer. Lastly, toluene with a +I group possesses little dipole moment. Thus the overall order is
-isomer. Lastly, toluene with a +I group possesses little dipole moment. Thus the overall order is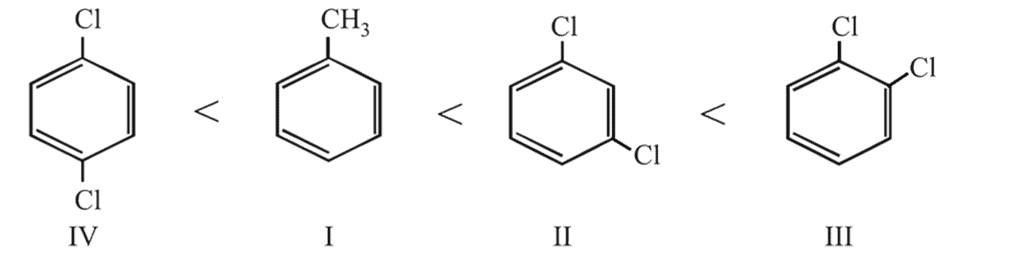
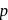 -nitrochlorobenzene as a major product. It further 4-nitrophenol.
-nitrochlorobenzene as a major product. It further 4-nitrophenol.
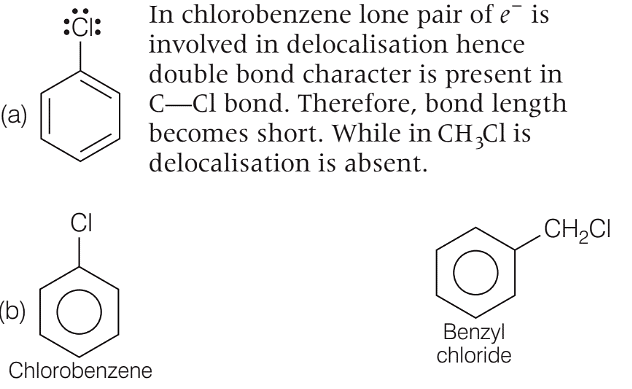
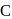 and
and 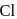 . So, it is difficult to replace form than form benzyl chloride.
. So, it is difficult to replace form than form benzyl chloride.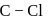 has double bond character in chlorobenzene.
has double bond character in chlorobenzene.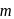 -dichlorobenzene due to
-dichlorobenzene due to 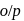 -directing effect of
-directing effect of 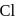 .
.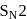 reaction (substitution nucleophilic bimolecular reaction) that involves an exchange of one halogen atom for another. It is an organic reaction, which uses an alkyl halide exchange into another alkyl halide through a reaction, wherein the metal halide is used.
reaction (substitution nucleophilic bimolecular reaction) that involves an exchange of one halogen atom for another. It is an organic reaction, which uses an alkyl halide exchange into another alkyl halide through a reaction, wherein the metal halide is used.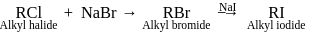
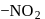 group withdraws electrons from
group withdraws electrons from 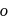 - and
- and 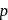 -positions and hence activates the
-positions and hence activates the 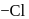 present on that position towards nucleophilic substitution.
present on that position towards nucleophilic substitution.