Preparation and Properties of Monohalides - Free MCQ Practice Test
MCQ Practice Test & Solutions: Test: Preparation and Properties of Monohalides (20 Questions)
You can prepare effectively for JEE Chemistry for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Preparation and Properties of Monohalides". These 20 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 20 minutes
- - Number of Questions: 20
Sign up on EduRev for free to attempt this test and track your preparation progress.
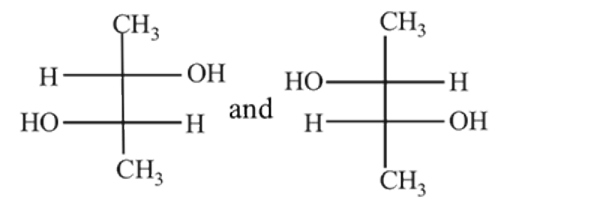
Which of the following pairs of compounds are enantiomers?
Detailed Solution: Question 1
The correct order of SN2 E2 ration for the % yield of product of the following halide is
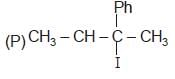
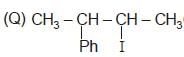
(R) CH3 — CH2 — I
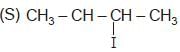
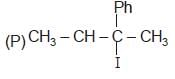
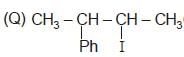
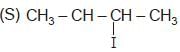
Detailed Solution: Question 2
Among the following organic halides in the increasing order of their dehydrohalogenation reactions in the presence of alcoholic 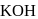
(A) 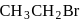
(B) 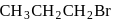
(C) 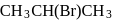
(D) 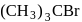
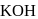
(A)
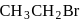
(B)
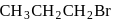
(C)
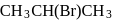
(D)
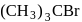
Detailed Solution: Question 3
Which one of the following is more reactive towards 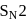 reaction?
reaction?
Detailed Solution: Question 4
Benzene reacts with 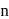 -propyl chloride in the presence of anhydrous
-propyl chloride in the presence of anhydrous 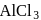 to give
to give
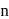 -propyl chloride in the presence of anhydrous
-propyl chloride in the presence of anhydrous 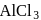 to give
to giveDetailed Solution: Question 5
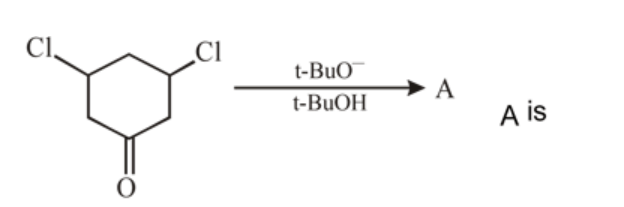
What is the product of the following reaction ?
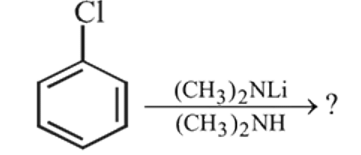
Detailed Solution: Question 6
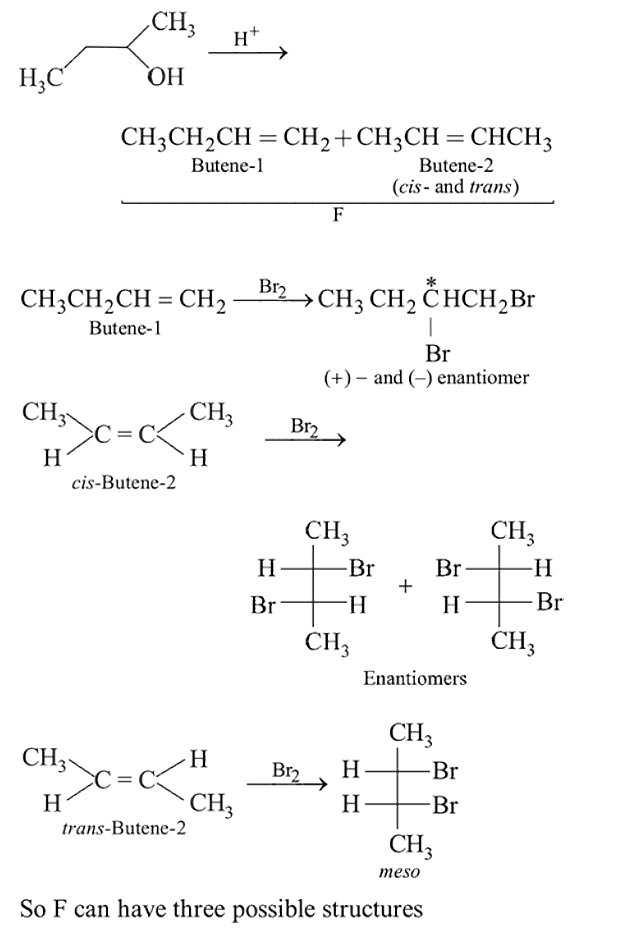
Identify 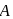 and
and 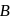 in the following reaction
in the following reaction
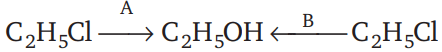
Detailed Solution: Question 7
Consider the reactions
(i) 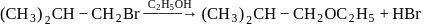
(ii) 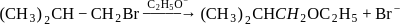
The mechanism of reactions (i) & (ii) are respectively
Detailed Solution: Question 8
How many stable alkenes on reaction with HCl will produce
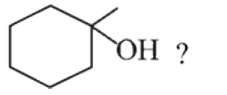
Detailed Solution: Question 9
Number of ambidentate nucleophiles among the following 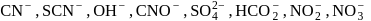
Detailed Solution: Question 10
Number of substrates which will show rearrangement is
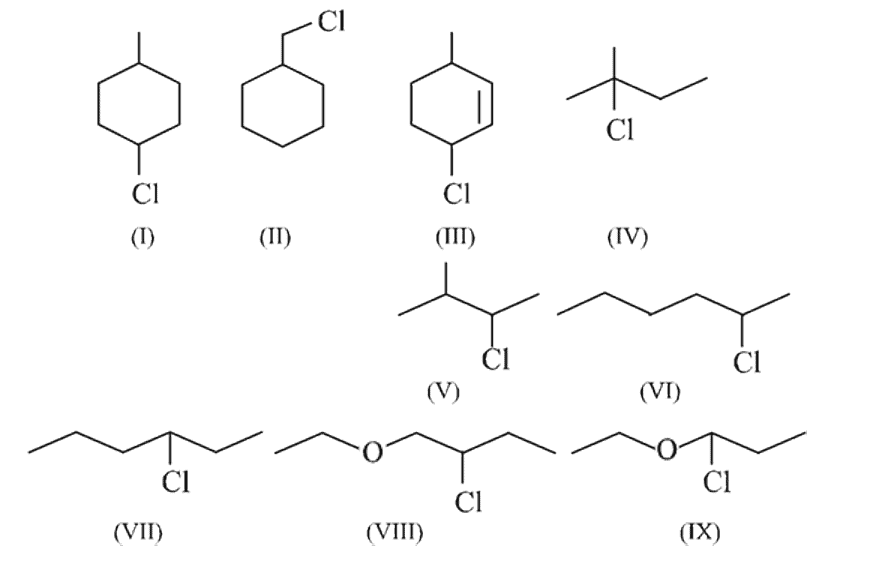
Detailed Solution: Question 11
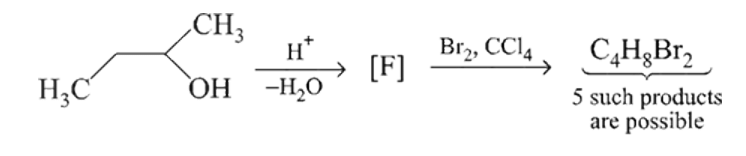
How many structures for 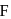 are possible?
are possible?
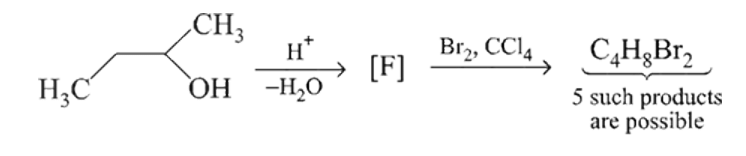
How many structures for
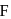 are possible?
are possible?Detailed Solution: Question 12
Which one of the following compounds most readily undrgoes substitution by 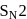 mechanism?
mechanism?
Detailed Solution: Question 13
Which of the following is fast debrominated?
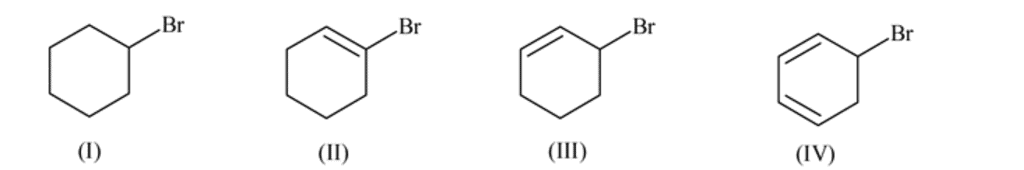
Detailed Solution: Question 14
The organic compound (P) of the reaction:
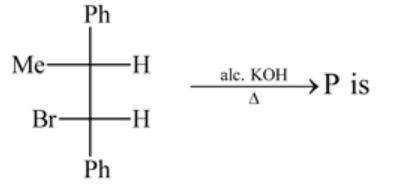
is

is
Detailed Solution: Question 15
Detailed Solution: Question 16
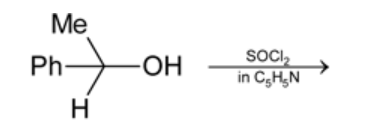
Which statement is true for the above reaction?

Which statement is true for the above reaction?
Detailed Solution: Question 17
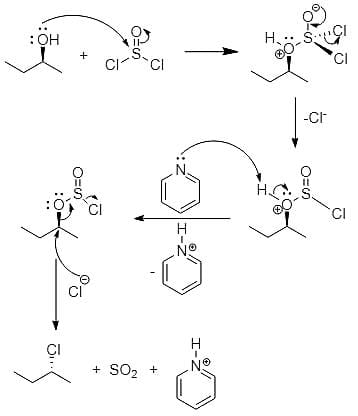
On on heating diethyl ether with conc- HI, 2 moles of which of the following is formed
Detailed Solution: Question 18
Alkyl halide reacts with an alcoholic solution of ammonia to give a mixture of:
Detailed Solution: Question 19
Product-I  Product-II The correct statement is
Product-II The correct statement is
Detailed Solution: Question 20
335 videos|699 docs|300 tests |



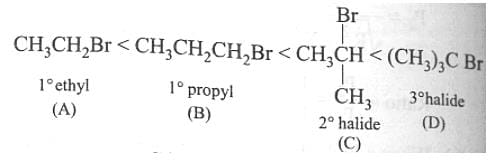
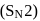 reaction involves reaction between reactant and nucleophile simultaneously without the formation of carbocation. Less hindered species are more reactive towards
reaction involves reaction between reactant and nucleophile simultaneously without the formation of carbocation. Less hindered species are more reactive towards 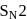 reaction. As
reaction. As  is least hindered alkyl halide, it will show more fast reaction towards
is least hindered alkyl halide, it will show more fast reaction towards 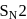 reaction.
reaction.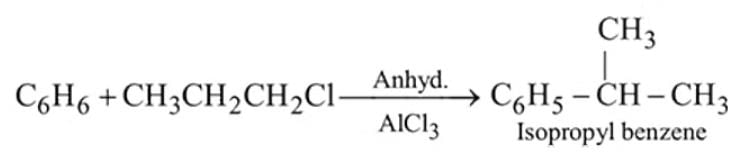
 .
.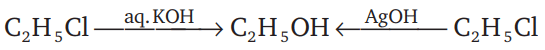
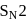 and
and 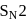
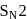 mechanism.
mechanism.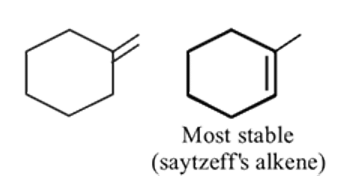
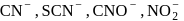
 mechanism is tert
mechanism is tert 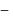 alkyl halide
alkyl halide  sec alkyl halide
sec alkyl halide  primary alkyl halide.
primary alkyl halide.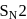 mechanism.
mechanism.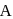 is also primary alkyl halide but in it, the carbon attached to is more hindered.
is also primary alkyl halide but in it, the carbon attached to is more hindered.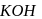 undergoes elimination reactions. For example:
undergoes elimination reactions. For example: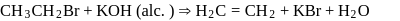
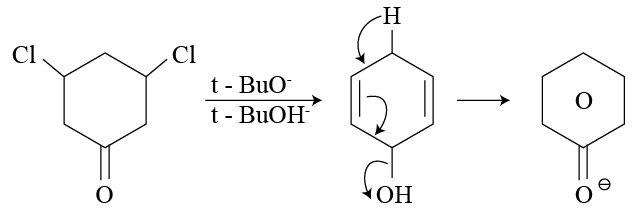
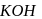 solution that functions as solvent gives alkoxide ions that act as a strong base. This base abstracts
solution that functions as solvent gives alkoxide ions that act as a strong base. This base abstracts 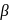 -Hydrogen atom from saturated substrate - alkyl halide.
-Hydrogen atom from saturated substrate - alkyl halide.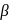 -Hydrogen atom is then transferred to the alkyl part to form an alkane and simultaneously a molecule of
-Hydrogen atom is then transferred to the alkyl part to form an alkane and simultaneously a molecule of 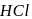 is eliminated.
is eliminated.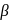 -hydrogen atoms of alkyl chloride to eliminate
-hydrogen atoms of alkyl chloride to eliminate  and form alkene.
and form alkene.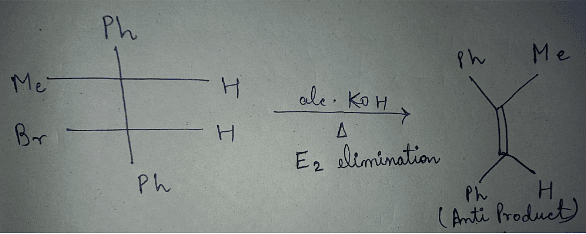
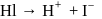
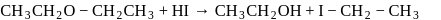
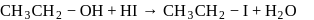

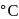
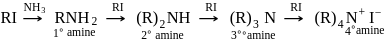
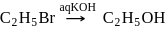 (Product-I)
(Product-I)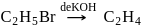 (Product-II)
(Product-II)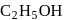 (Product-I) isomer is
(Product-I) isomer is 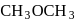
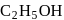 denydration
denydration