Preparation and Properties of colloids - Free MCQ Practice Test with solutions,
MCQ Practice Test & Solutions: Test: Preparation and Properties of colloids (15 Questions)
You can prepare effectively for JEE Chemistry for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Preparation and Properties of colloids". These 15 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 20 minutes
- - Number of Questions: 15
Sign up on EduRev for free to attempt this test and track your preparation progress.
During the action of enzyme "Zymase", glucose is converted into_____, with the liberation of carbon dioxide gas.
Detailed Solution: Question 1
Detailed Solution: Question 2
The efficiency of an enzyme in catalysing a reaction is due to its capacity
Detailed Solution: Question 3
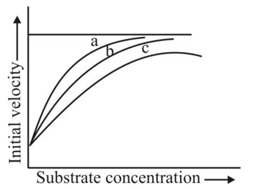
The figure given below shows three velocity-substrate concentration curves for an enzyme reaction. What do the curves 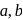 and c depict respectively?
and c depict respectively?
Detailed Solution: Question 4
The density of gold is 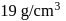 . If
. If 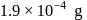 of gold is dispersed in one litre of water to give a sol having spherical gold particles of radius 10
of gold is dispersed in one litre of water to give a sol having spherical gold particles of radius 10 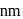 , then the number of gold particles per
, then the number of gold particles per 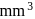 of
of
the sol will be :
Detailed Solution: Question 5
Detailed Solution: Question 6
Cloud or fog is a colloidal system in which the dispersed phase and the dispersion medium are
Detailed Solution: Question 7
Which of the following constitutes irreversible colloidal system in water as dispersion medium?
Detailed Solution: Question 8
Which one of the following statements is not correct in respect of lyophilic sols?
Detailed Solution: Question 9
Detailed Solution: Question 10
The stabilization of the dispersed phase in a lyophobic sol is due to
Detailed Solution: Question 11
A detergent 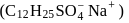 solution becomes a colloidal sol at a concentration of
solution becomes a colloidal sol at a concentration of 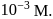 On an average
On an average 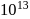 colloidal particles are present in
colloidal particles are present in 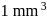 . What is the average number of ions which are contained by one colloidal particle (micelle)?
. What is the average number of ions which are contained by one colloidal particle (micelle)?  Given:
Given: 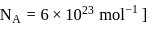
Detailed Solution: Question 12
Detailed Solution: Question 13
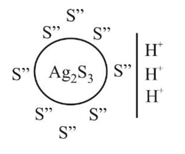
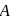 white precipitate of
white precipitate of 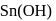 is peptized with di.
is peptized with di. 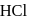 . The sol particle will carry
. The sol particle will carry
Detailed Solution: Question 14
The sol prepared by Bredig's Arc method is X and the charge of sol particles of it is q. 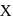 and q are respectively.
and q are respectively.
Detailed Solution: Question 15
335 videos|697 docs|300 tests |


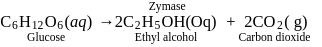

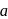 shows normal enzyme reaction, while curve
shows normal enzyme reaction, while curve 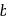 shows a competitive inhibition reaction, in which competitive inhibitors that resemble the substrate molecules, bind to the active site of the enzyme, whereas, curve
shows a competitive inhibition reaction, in which competitive inhibitors that resemble the substrate molecules, bind to the active site of the enzyme, whereas, curve 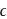 shows non-competitive inhibition reaction, in which the inhibitor binds to a part of the enzyme or enzyme substrate complex, other than the active site, known as the allosteric site.
shows non-competitive inhibition reaction, in which the inhibitor binds to a part of the enzyme or enzyme substrate complex, other than the active site, known as the allosteric site.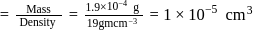
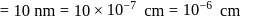
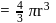
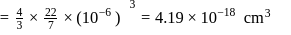
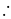 No. of gold sol particles in
No. of gold sol particles in 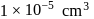
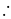 No. of gold sol particles in one
No. of gold sol particles in one 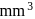
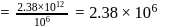
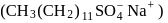 in 1 litre solution
in 1 litre solution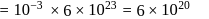
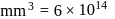
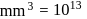 No. of molecules per colloidal particle
No. of molecules per colloidal particle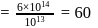
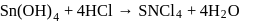
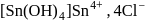 (Positive charge)
(Positive charge)