Preparation and Reactions of Glucose - Free MCQ Practice Test with solutions,
MCQ Practice Test & Solutions: Test: Preparation and Reactions of Glucose (15 Questions)
You can prepare effectively for JEE Chemistry for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Preparation and Reactions of Glucose ". These 15 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 20 minutes
- - Number of Questions: 15
Sign up on EduRev for free to attempt this test and track your preparation progress.
Glucose molecule reacts with 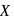 number of molecules of phenylhydrazine to yield osazone. The value of
number of molecules of phenylhydrazine to yield osazone. The value of 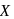 is
is
Detailed Solution: Question 1
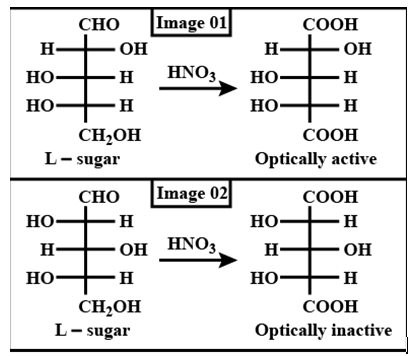
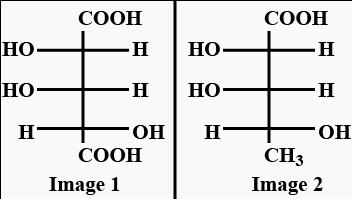
Which L-sugar on oxidation gives an optically active dibasic acid ( 2 COOH groups)?
Detailed Solution: Question 2
In cells the net production of ATP molecules generated from one glucose molecule is
Detailed Solution: Question 3
Hydrolysis of sucrose with dilute aqueous sulphuric acid yields
Detailed Solution: Question 4
In osazone formation, glucose reacts with three molecules of phenylhydrazine. Which statement is true regarding this?
Detailed Solution: Question 5
What will happen when 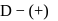 -glucose is treated with methanolic
-glucose is treated with methanolic 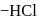 followed by Tollens' reagent?
followed by Tollens' reagent?
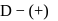 -glucose is treated with methanolic
-glucose is treated with methanolic 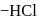 followed by Tollens' reagent?
followed by Tollens' reagent?Detailed Solution: Question 6
Among the three compounds shown below, two yield the same product on reaction with warm 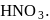 The exception is:
The exception is:
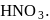 The exception is:
The exception is:Detailed Solution: Question 7
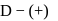 -Glucose
-Glucose 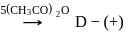 -Glucose pentaacetate
-Glucose pentaacetate
Which statement is true about glucose pentaacetate?
Detailed Solution: Question 8
The two forms of D-glucopyranose obtained from the solution of D-glucose are called
Detailed Solution: Question 9
The number of molecules of ATP produced per molecule of glucose in glycolysis is
Detailed Solution: Question 10
Reducing saccharides among the following are
Detailed Solution: Question 11
Carbohydrates are stored in plants and animals in which of the following forms respectively?
Detailed Solution: Question 12
Which of the following statements are true about carbohydrates?
(i) Monosaccharides can be hydrolysed.
(ii) The two monosaccharide units obtained on hydrolysis of a disaccharide can either be same or different.
(iii) Polysaccharides are now sweet in taste.
(iv) All monosaccharides are not reducing. sugars.
(i) Monosaccharides can be hydrolysed.
(ii) The two monosaccharide units obtained on hydrolysis of a disaccharide can either be same or different.
(iii) Polysaccharides are now sweet in taste.
(iv) All monosaccharides are not reducing. sugars.
Detailed Solution: Question 13
Which of the following does not reduce Benedict's solution?
Detailed Solution: Question 14
Detailed Solution: Question 15
335 videos|699 docs|300 tests |


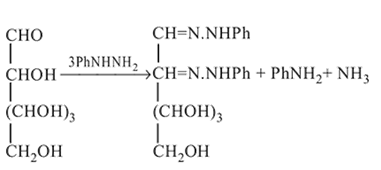

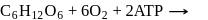
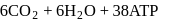
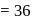
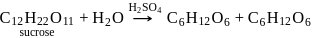
 in fructose) group. Second molecule of the reagent oxidizes
in fructose) group. Second molecule of the reagent oxidizes  - at position 2 (in aldoses) or
- at position 2 (in aldoses) or  at position 1 (in ketoses) to form
at position 1 (in ketoses) to form 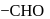 or
or 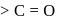 respectively. The third molecule again undergoes nucleophilic addition on the newly developed carbonyl group to form osazone.
respectively. The third molecule again undergoes nucleophilic addition on the newly developed carbonyl group to form osazone.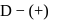 - glucose is treated with methanolic
- glucose is treated with methanolic 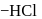 followed by Tollens' reagent, no characteristic colour or ppt. will be formed.
followed by Tollens' reagent, no characteristic colour or ppt. will be formed.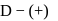 glucose with methanolic
glucose with methanolic 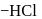 leads to formation of methyl glucoside
leads to formation of methyl glucoside of hemiacetal is methylated to form acetal). Acetal, is not hydrolysable by base, so it will not respond Tollen's reagent.
of hemiacetal is methylated to form acetal). Acetal, is not hydrolysable by base, so it will not respond Tollen's reagent.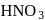 oxidation of aldehyde and alcohol takes place which gives carboxylic acid.
oxidation of aldehyde and alcohol takes place which gives carboxylic acid.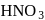 . Thus (a)
. Thus (a) 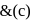 will give same product i.e.
will give same product i.e.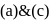 .
.
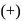 -glucose, it is the
-glucose, it is the 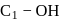 of the hemiacetal that is acetylated and not the
of the hemiacetal that is acetylated and not the 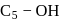 that forms the ring (cyclic structure). Since equilibrium with the open-chain aldehyde is prevented, the penta-acetate does not respond the aldehydic reactions.
that forms the ring (cyclic structure). Since equilibrium with the open-chain aldehyde is prevented, the penta-acetate does not respond the aldehydic reactions.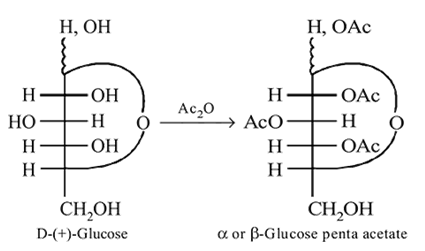
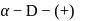 -glucopyranose and
-glucopyranose and 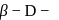 (+)-glucopyranose. These are anomers (a pair of stereoisomers) which differ in configuration only around first carbon atom are called anomers.
(+)-glucopyranose. These are anomers (a pair of stereoisomers) which differ in configuration only around first carbon atom are called anomers.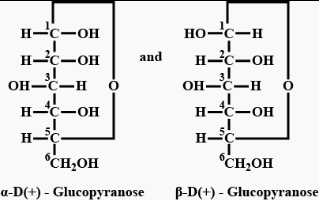
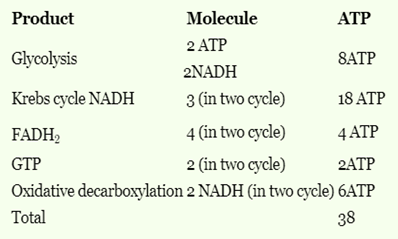
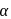 -hydroxy ketonic group, which is also reducing group (different from ordinary ketonic group)
-hydroxy ketonic group, which is also reducing group (different from ordinary ketonic group)