Properties and Preparation of Alkynes - Free MCQ Practice Test with solutions,
MCQ Practice Test & Solutions: Test: Properties and Preparation of Alkynes (10 Questions)
You can prepare effectively for JEE Weekly Tests for JEE Preparation with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Properties and Preparation of Alkynes". These 10 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 20 minutes
- - Number of Questions: 10
Sign up on EduRev for free to attempt this test and track your preparation progress.
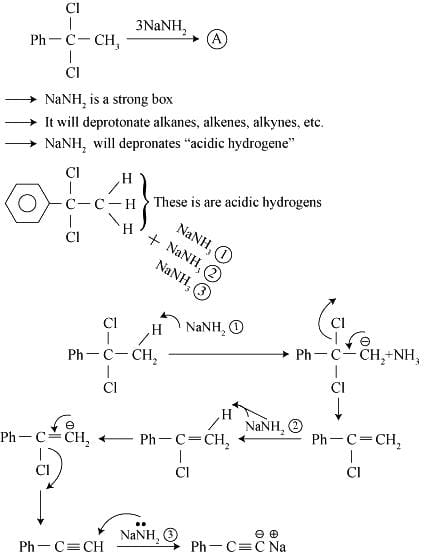
Detailed Solution: Question 1
End product of the following sequence of reactions is
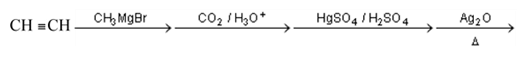
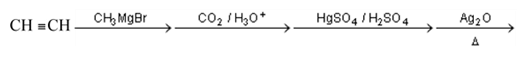
Detailed Solution: Question 2
A compound (X) 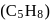 reacts with ammonical
reacts with ammonical 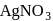 to give a white precipitate, and on oxidation with hot alkaline
to give a white precipitate, and on oxidation with hot alkaline 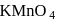 gives the acid,
gives the acid,  , therefore
, therefore 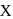 is
is 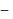
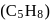 reacts with ammonical
reacts with ammonical 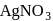 to give a white precipitate, and on oxidation with hot alkaline
to give a white precipitate, and on oxidation with hot alkaline 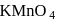 gives the acid,
gives the acid,  , therefore
, therefore 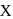 is
is 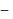
Detailed Solution: Question 3
An unknown compound A has a molecular formula 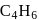 , when
, when  is treated with an excess of
is treated with an excess of 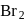 , a new substance
, a new substance 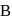 with formula
with formula 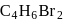 is formed. A forms a white precipitate with ammonical silver nitrate solution. A may be
is formed. A forms a white precipitate with ammonical silver nitrate solution. A may be
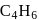 , when
, when 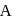 is treated with an excess of
is treated with an excess of 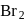 , a new substance
, a new substance 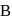 with formula
with formula 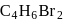 is formed. A forms a white precipitate with ammonical silver nitrate solution. A may be
is formed. A forms a white precipitate with ammonical silver nitrate solution. A may beDetailed Solution: Question 4
CH3 − C ≡ CMgBr can be prepared by the reaction of
Detailed Solution: Question 5
Detailed Solution: Question 6
The major product formed in the following reaction is 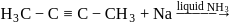
Detailed Solution: Question 7
Detailed Solution: Question 8
Identify the reagent from the following list which can easily distinguish between 1 -butyne and 2-butyne
Detailed Solution: Question 9
Detailed Solution: Question 10


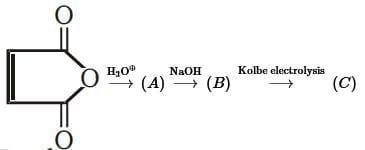
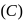 of above reaction is:
of above reaction is: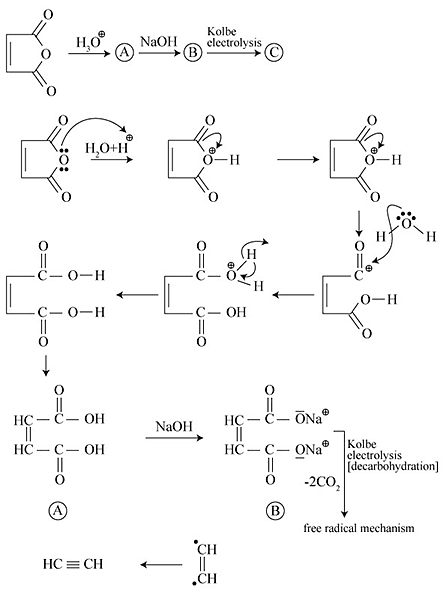
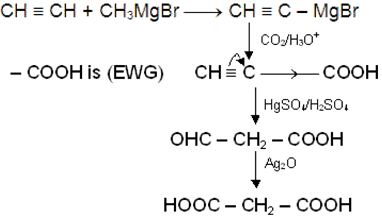

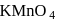 is:
is: 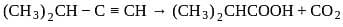
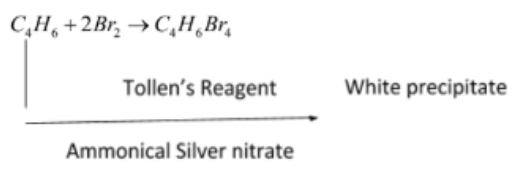
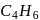 is butyne. It can either be But-1-yne or But-2-yne.
is butyne. It can either be But-1-yne or But-2-yne.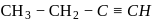 But-1-yne
But-1-yne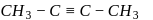 But-2-yne
But-2-yne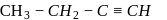 is acidic in nature.
is acidic in nature.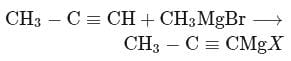
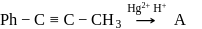
 is:
is: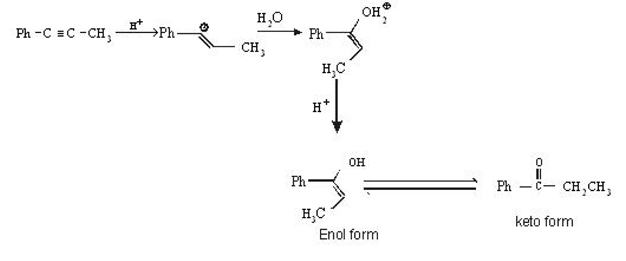
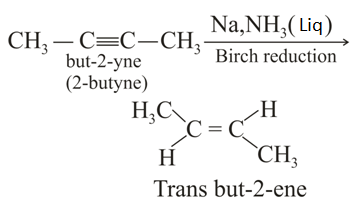
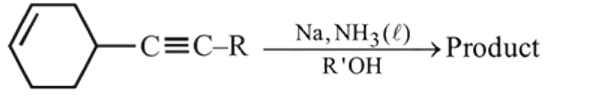
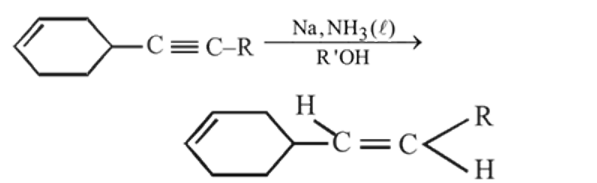
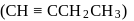
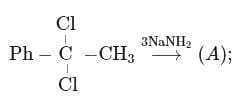 Product
Product 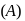 is : Product
is : Product