Purification of Organic Compounds - Free MCQ Practice Test with solutions,
MCQ Practice Test & Solutions: Test: Purification of Organic Compounds (20 Questions)
You can prepare effectively for JEE Chemistry for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Purification of Organic Compounds". These 20 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 20 minutes
- - Number of Questions: 20
Sign up on EduRev for free to attempt this test and track your preparation progress.
For the separation of two miscible liquids which method is used?
Detailed Solution: Question 1
Detailed Solution: Question 2
In Lassaigne's test, the organic compound is fused with a piece of sodium metal in order to
Detailed Solution: Question 3
A is a lighter phenol and B is an aromatic carboxylic acid. Separation of mixture of A and B can be carried out easily by using a solution of
Detailed Solution: Question 4
In Dumas method, 0.3 g of an organic compound gave 45 mL of nitrogen at STP. The percentage of nitrogen is
Detailed Solution: Question 5
The Lassaigne's extract is boiled with dil. HNO3 before testing for halogens because
Detailed Solution: Question 6
The best method for the separation of naphthalene and benzoic acid from their mixture is:
Detailed Solution: Question 7
Detailed Solution: Question 8
Beilstein test is used for determination of
Detailed Solution: Question 9
2.79 g of an organic compound when heated in Carius tube with conc. HNO3 and H3PO4 formed is converted into MgNH4.PO4 ppt. The ppt. on heating gave 1.332 g of Mg2P2O7. The percentage of P in the compound is
Detailed Solution: Question 10
If 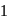 equivalent
equivalent 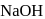 reacts with the given compound:
reacts with the given compound:
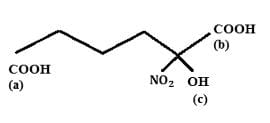
Choose the correct option:
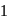 equivalent
equivalent  reacts with the given compound:
reacts with the given compound:
Detailed Solution: Question 11
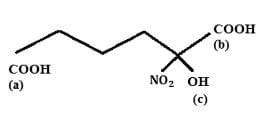
Find out the double bond equivalent (DBE) value of the given following compound:
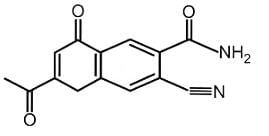

Detailed Solution: Question 12
The prussian blue colour obtained during the test of nitrogen by Lassaigne's test is due to the formation of :
Detailed Solution: Question 13
The purification of naphthalene can be done by
Detailed Solution: Question 14
Two substances having slightly different solubility can be separated by
Detailed Solution: Question 15
Which of the following statements is true regarding the fractional distillation of a mixture of two liquids?
Detailed Solution: Question 16
A compound which decomposes before its boiling point can be purified by the method of
Detailed Solution: Question 17
Detailed Solution: Question 18
Detailed Solution: Question 19
Chromatography is based on the phenomenon of
Detailed Solution: Question 20
335 videos|699 docs|300 tests |



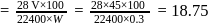
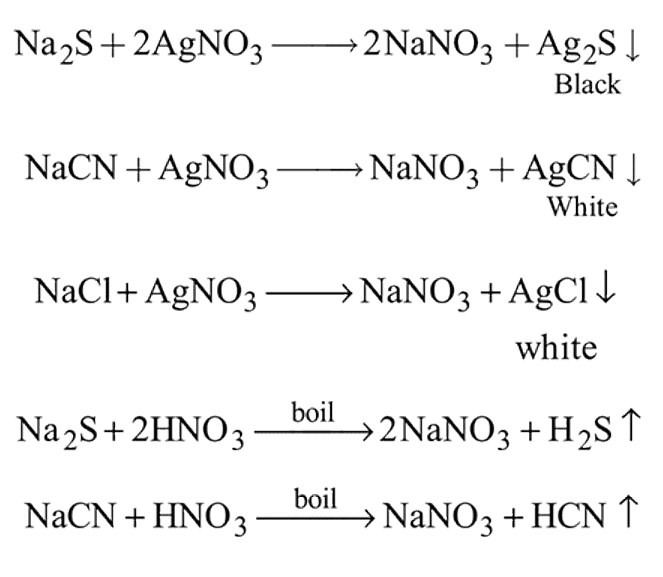
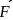
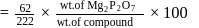
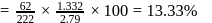
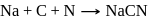
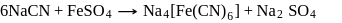
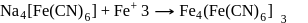
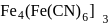 is responsible for prussian blue colour obtained during the test of Nitrogen and Lassaigne test. O-nitrophenal is more volatile than pnitrophenal. There is intramolecular H - bonding in O- nitrophenal while in P- nitrophenal there is intermolecular hydrogen bonding which cause association of molecule. Intermolecular H - bondingincrease, boiling point increases while volatility reduces.
is responsible for prussian blue colour obtained during the test of Nitrogen and Lassaigne test. O-nitrophenal is more volatile than pnitrophenal. There is intramolecular H - bonding in O- nitrophenal while in P- nitrophenal there is intermolecular hydrogen bonding which cause association of molecule. Intermolecular H - bondingincrease, boiling point increases while volatility reduces.