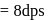Rate of reactions - Free MCQ Practice Test with solutions, JEE Chemistry
MCQ Practice Test & Solutions: Test: Rate of reactions (15 Questions)
You can prepare effectively for JEE Chemistry for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Rate of reactions". These 15 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 20 minutes
- - Number of Questions: 15
Sign up on EduRev for free to attempt this test and track your preparation progress.
The reaction rate is defined as the rate at which the concentration of the reactants __________ with time or the concentration of products ___________ with time.
Detailed Solution: Question 1
Consider the following reaction
A⟶ Products
This reaction is completed in 100 min. The rate constant of this reaction at t1 = 10 min, is 10−2 min−1. What is the rate constant (in min−1) at t2 = 20 min?
A⟶ Products
This reaction is completed in 100 min. The rate constant of this reaction at t1 = 10 min, is 10−2 min−1. What is the rate constant (in min−1) at t2 = 20 min?
Detailed Solution: Question 2
Which of the following terms would not be determined experimentally?
Detailed Solution: Question 3
Consider the reaction
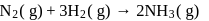
The equality relationship between 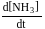 and
and 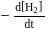 is
is
Detailed Solution: Question 4
A following mechanism has been proposed for reaction 2 A + B → D + E
A + B → C + D(slow)
A + C → E (fast)
The rate law expression for the reaction is:
A + B → C + D(slow)
A + C → E (fast)
The rate law expression for the reaction is:
Detailed Solution: Question 5
The rate law for the reaction 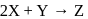 is Rate
is Rate 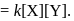 The correct statement with regard to this relation is
The correct statement with regard to this relation is
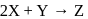 is Rate
is Rate 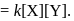 The correct statement with regard to this relation is
The correct statement with regard to this relation isDetailed Solution: Question 6
In a reaction, 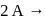 products, the concentration of
products, the concentration of 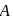 decreases from
decreases from 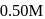 to
to 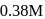 in
in 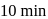 . What is the rate of disappearance of A (in
. What is the rate of disappearance of A (in 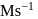 ) during this interval?
) during this interval?
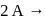 products, the concentration of
products, the concentration of 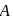 decreases from
decreases from 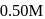 to
to 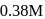 in
in 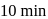 . What is the rate of disappearance of A (in
. What is the rate of disappearance of A (in 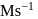 ) during this interval?
) during this interval?Detailed Solution: Question 7
Detailed Solution: Question 8
Detailed Solution: Question 9
The inversion of cane sugar proceeds with half-life of 500 minute at 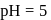 for any concentration of sugar. However if
for any concentration of sugar. However if 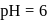 , the half-life changes to 50 minute. The rate law expression for the sugar inversion can be written as
, the half-life changes to 50 minute. The rate law expression for the sugar inversion can be written as
Detailed Solution: Question 10
Reaction rates are determined by all of the following factors except:
Detailed Solution: Question 11
For a reaction 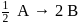 , rate of disappearance of '
, rate of disappearance of ' 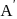 is related to the rate of appearance of 'B' by the expression
is related to the rate of appearance of 'B' by the expression
Detailed Solution: Question 12
The terms rate of reaction and rate of appearance (or disappearance) of reactant (or product)
Detailed Solution: Question 13
For the reaction 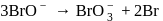 in alkaline aqueous solution, the value of the second order (in
in alkaline aqueous solution, the value of the second order (in 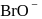 ) rate constant at
) rate constant at 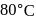 in the rate law
in the rate law 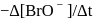 was found to be
was found to be
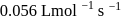 . Rate constant when the rate law is written for
. Rate constant when the rate law is written for 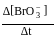 is
is
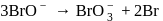 in alkaline aqueous solution, the value of the second order (in
in alkaline aqueous solution, the value of the second order (in 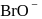 ) rate constant at
) rate constant at 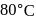 in the rate law
in the rate law 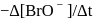 was found to be
was found to be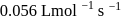 . Rate constant when the rate law is written for
. Rate constant when the rate law is written for 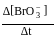 is
isDetailed Solution: Question 14
A G.M. counter is used to study the radioactive process of first-order. In the absence of radioactive substance 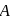 , it counts 3 disintegration per second (dps). When
, it counts 3 disintegration per second (dps). When 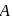 is placed in the G.M. counter, it records 23 dps at the start and 13 dps after 10 minutes. It records
is placed in the G.M. counter, it records 23 dps at the start and 13 dps after 10 minutes. It records 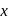 dps after next 10 minutes and
dps after next 10 minutes and 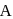 has half-life period y minutes.
has half-life period y minutes. 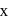 and
and 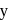 are
are
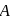 , it counts 3 disintegration per second (dps). When
, it counts 3 disintegration per second (dps). When 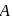 is placed in the G.M. counter, it records 23 dps at the start and 13 dps after 10 minutes. It records
is placed in the G.M. counter, it records 23 dps at the start and 13 dps after 10 minutes. It records 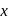 dps after next 10 minutes and
dps after next 10 minutes and 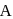 has half-life period y minutes.
has half-life period y minutes. 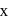 and
and 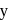 are
areDetailed Solution: Question 15
335 videos|699 docs|300 tests |


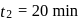 also
also 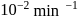 (constant).
(constant).
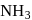 and
and 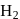 , then
, then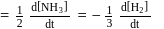
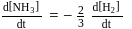
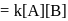
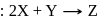
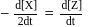
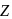 is half of the rate of disappearance of
is half of the rate of disappearance of 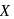 .
.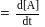
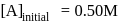
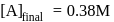
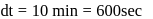
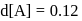
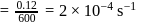 .
.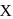 Hence X
Hence X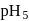 , the half-life is independent of the concentration of sugar. Hence, the reaction is of first order in sugar.
, the half-life is independent of the concentration of sugar. Hence, the reaction is of first order in sugar.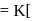 sugar
sugar 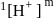 .
.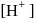 is
is 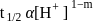
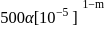
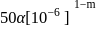
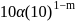
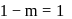
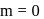
 .
.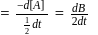 or
or 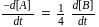
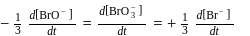
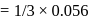
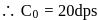
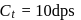
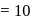 minutes
minutes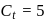 dps
dps