Refining & Reduction of Crude Metal, Flux and Refractory materials - Free
MCQ Practice Test & Solutions: Test: Refining & Reduction of Crude Metal, Flux and Refractory materials (15 Questions)
You can prepare effectively for JEE Chemistry for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Refining & Reduction of Crude Metal, Flux and Refractory materials". These 15 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 20 minutes
- - Number of Questions: 15
Sign up on EduRev for free to attempt this test and track your preparation progress.
Which of the following statements regarding electrolytic refining of copper is incorrect?
Detailed Solution: Question 1
Which one of the following ores is concentrated by chemical leaching method?
Detailed Solution: Question 2
If an impurity in a metal has a greater affinity for oxygen and is more easily oxidised than the metal, then the purification of metal may be carried out by
Detailed Solution: Question 3
van Arkel method of purification of metals involves converting the metal to a
Detailed Solution: Question 4
Which of the following pairs of metals is purified by van Arkel method?
Detailed Solution: Question 5
Which of the following processes involve the roasting process?
Detailed Solution: Question 6
In electrorefining of copper some gold is deposited as
Detailed Solution: Question 7
In electro-refining of metal the impure metal is made the anode and a strip of pure metal, the cathode, during the electrolysis of an aqueous solution of a complex metal salt. This method cannot be used for refining of
Detailed Solution: Question 8
Match the following items of List-I with those of List-II
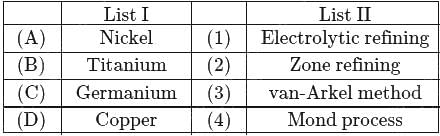
The correct answer is

The correct answer is
Detailed Solution: Question 9
The metal purified by. Mond process is 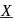 and metal purified by van Arkel method is
and metal purified by van Arkel method is 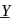 . X and Y respectively are
. X and Y respectively are
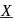 and metal purified by van Arkel method is
and metal purified by van Arkel method is 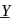 . X and Y respectively are
. X and Y respectively areDetailed Solution: Question 10
During the electrolysis of cryolite, aluminium and fluorine are formed in ........ molar ratio :
Detailed Solution: Question 11
Which method of purification is represented by the following equation?
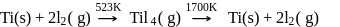
Detailed Solution: Question 12
Refractory materials are generally used in furnaces because
Detailed Solution: Question 13
Which one of the following ores is not concentrated by froth floatation process?
Detailed Solution: Question 14
Which one of the following is used as an acid flux in metallurgy?
Detailed Solution: Question 15
332 videos|699 docs|300 tests |



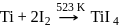 (stable compound)
(stable compound) 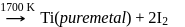
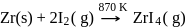
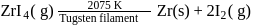
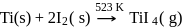



 Pure titanium
Pure titanium
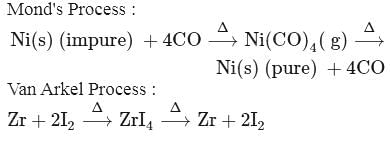
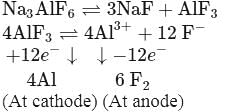
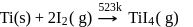
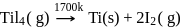
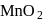 . Hence not concentrated by froth floatation process.
. Hence not concentrated by froth floatation process.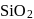 (silica) is used as an acid flux in metallurgy. It reacts with gangue to form slag.
(silica) is used as an acid flux in metallurgy. It reacts with gangue to form slag.