Stoichiometric and Non-stoichiometric defects - Free MCQ Practice Test
MCQ Practice Test & Solutions: Test: Stoichiometric and Non-stoichiometric defects (10 Questions)
You can prepare effectively for JEE Chemistry for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Stoichiometric and Non-stoichiometric defects". These 10 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 20 minutes
- - Number of Questions: 10
Sign up on EduRev for free to attempt this test and track your preparation progress.
When n-type and p-type semiconductors are allowed to come into contact
Detailed Solution: Question 1
Superconductors are derived from compounds of
Detailed Solution: Question 2
If we mix a pentavalent impurity in a crystal lattice of germanium, what type of semiconductor formation will occur?
Detailed Solution: Question 3
To get a 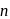 - type semiconductor, the impurity to be added to silicon should have which of the following number of valence electrons
- type semiconductor, the impurity to be added to silicon should have which of the following number of valence electrons
Detailed Solution: Question 4
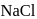 is heated in an atmosphere of sodium vapour. The resultant yellow colour is due to the formation of
is heated in an atmosphere of sodium vapour. The resultant yellow colour is due to the formation of
Detailed Solution: Question 5
Non stoichiometric defects are formed by
Detailed Solution: Question 6
What type of crystal defects is indicated in the diagram given below?


Detailed Solution: Question 7
Each of the following solids show, the Frenkel defect except
Detailed Solution: Question 8
Schottky defect in crystals is observed when
Detailed Solution: Question 9
Which one of the following has Frenkel defect
Detailed Solution: Question 10
335 videos|697 docs|300 tests |


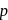 -type semiconductors are allowed to come into contact then some electrons will flow from
-type semiconductors are allowed to come into contact then some electrons will flow from 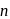 to
to 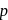 .
.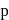 block elements.
block elements.
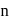 -type, since electron is set free.
-type, since electron is set free.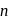 -type, impurity added to silicon should have more than 4 valence electrons.
-type, impurity added to silicon should have more than 4 valence electrons.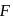 -centeres that give rise to intresting colour in alkali halides.
-centeres that give rise to intresting colour in alkali halides.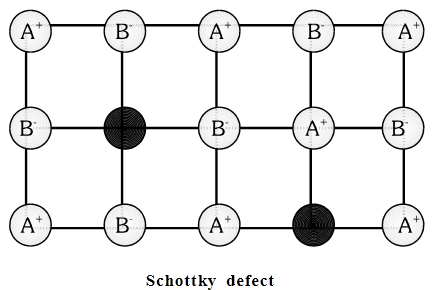
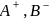 , equal number of cations and anions are missing from their lattice sites so that the electrical neutrality is maintained. The defect is called Schottky defect.
, equal number of cations and anions are missing from their lattice sites so that the electrical neutrality is maintained. The defect is called Schottky defect.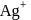 and
and 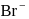 ions.
ions.