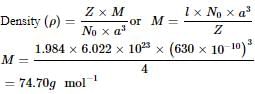The Solid State (Old NCERT) - Free MCQ Practice Test with solutions, JEE
MCQ Practice Test & Solutions: Test: The Solid State (Old NCERT) (25 Questions)
You can prepare effectively for JEE Chapter-wise Tests for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: The Solid State (Old NCERT)". These 25 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 35 minutes
- - Number of Questions: 25
Sign up on EduRev for free to attempt this test and track your preparation progress.
Substances that are strongly attracted by applied magnetic field and can be permanently magnetized are
Detailed Solution: Question 1
Which type of solid conduct electricity in molten state but not in solid state?
Detailed Solution: Question 2
Yellow colour of NaCl crystals in sodium vapour is due to
Detailed Solution: Question 3
Detailed Solution: Question 4
Detailed Solution: Question 5
Detailed Solution: Question 6
Solid X is a very hard electrical insulator in solid as well as in molten state. It melts at extremely high temperature. Solid X is a
Detailed Solution: Question 7
Nature of binding forces present in carbondioxide molecules in solid state are?
Detailed Solution: Question 8
What type of interaction hold the molecules together in a polar molecular solid?
Detailed Solution: Question 9
An atom located at the body center of a cubic unit cell is shared by
Detailed Solution: Question 10
Detailed Solution: Question 11
The coordination number of AB having rock salt geometry is
Detailed Solution: Question 12
MgO has the structure of NaCl. The coordination number of the ions in MgO is
Detailed Solution: Question 13
Detailed Solution: Question 14
A metal crystallizes with a face-centered cubic lattice. The edge of the unit cell is 408 pm. The diameter of the metal atom is
Detailed Solution: Question 15
An element forms a cubic unit cell with edge length 405 pm. Molar mass of this element is 2.7 X 10-2 Kg/mol and its density is given as 2.7 X 103 Kg/m3. How many atoms of this elements are present per unit cell.
Detailed Solution: Question 16
A group of 14 element is converted into n – type semiconductor by dopping it with
Detailed Solution: Question 17
Group 14 element is converted to p – type semiconductor by dopping it with
Detailed Solution: Question 18
Detailed Solution: Question 19
A substance forms face centered cubic crystals. Its density is 1.984 g/cm3 and the length of the edge of the unit cell is 630 pm. Calculate the molar mass in g/mol?
Detailed Solution: Question 20
Detailed Solution: Question 21
Detailed Solution: Question 22
In n – type semiconductor current is carried by
Detailed Solution: Question 23
Detailed Solution: Question 24
Which transition metal oxide has appearance and conductivity like that of copper?
Detailed Solution: Question 25
446 docs|929 tests |