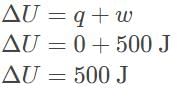Thermodynamics - Free MCQ Practice Test with solutions, MCAT General Chemistry
MCQ Practice Test & Solutions: Test: Thermodynamics (10 Questions)
You can prepare effectively for MCAT General Chemistry for MCAT with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Thermodynamics". These 10 questions have been designed by the experts with the latest curriculum of MCAT 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 10 minutes
- - Number of Questions: 10
Sign up on EduRev for free to attempt this test and track your preparation progress.
When heating a solution, a scientist detects a temperature increase in the solution during a period of time. Which of the following statements accurately characterizes the solution during this period?
Detailed Solution: Question 1
Equal amounts of heat are absorbed by 100 g, samples of various solid metals with differing specific heat values. Which of the following statements is true regarding metals and their specific heat values?
Detailed Solution: Question 2
In a system undergoing adiabatic compression, what are the values of internal energy and heat if work done on the system is 500 J?
Detailed Solution: Question 3
Additional gas is pumped inside a rigid container that stores compressed gas. Which of the following is a true statement about this system?
Detailed Solution: Question 4
Which of the following scenarios violates the first law of thermodynamics, “the conservation of energy?"
Detailed Solution: Question 5
A hot object is placed next to a cold object so that they are touching. Which of the following statements is true?
I. Heat will transfer from the hot object to the cold object because the hot object has a higher temperature.
II. The two objects are in thermal equilibrium
III. Internal energy will transfer from the hot object to the cold object because the hot object has greater internal energy.
I. Heat will transfer from the hot object to the cold object because the hot object has a higher temperature.
II. The two objects are in thermal equilibrium
III. Internal energy will transfer from the hot object to the cold object because the hot object has greater internal energy.
Detailed Solution: Question 6
Atmospheric gases absorb more energy than they emit. If we consider a gas to be a closed system, which of the following is true?
Detailed Solution: Question 7
What is the net work done on the gas as it goes from point A to B on the Pressure vs Volume diagram?
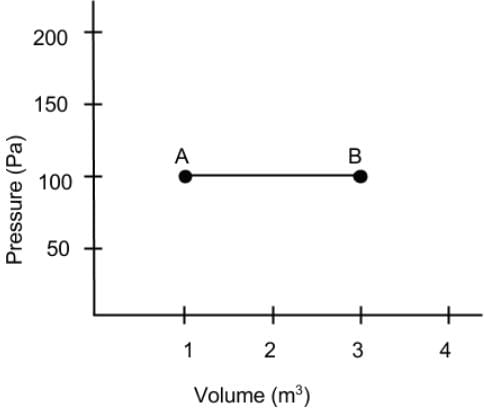

Detailed Solution: Question 8
What is the net work done on the gas as it goes from point C to D and then to E on the Pressure vs Volume diagram?
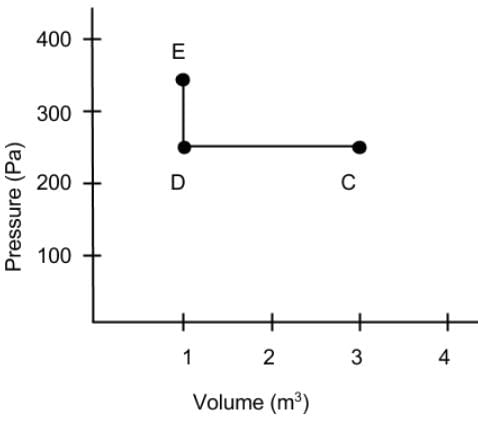

Detailed Solution: Question 9
Detailed Solution: Question 10
164 videos|15 docs|16 tests |