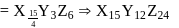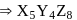Types of Solids and Properties - Free MCQ Practice Test with solutions,
MCQ Practice Test & Solutions: Test: Types of Solids and Properties (10 Questions)
You can prepare effectively for JEE Chemistry for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Types of Solids and Properties". These 10 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 20 minutes
- - Number of Questions: 10
Sign up on EduRev for free to attempt this test and track your preparation progress.
Detailed Solution: Question 1
Which of the following is not a crystalline solid?
Detailed Solution: Question 2
Which one of the following forms a molecular solid when solidified?
Detailed Solution: Question 3
Detailed Solution: Question 4
What is the energy gap between valence band and conduction band in crystal of insulators?
Detailed Solution: Question 5
In a solid 'AB' having the NaCl structure, 'A' atoms occupy the corners in cubic unit cell. If all the face-centered atoms along one of the axes are removed, then the resultant stoichiometry of the solid is
Detailed Solution: Question 6
Assertion (A): White tin is an example of tetragonal system.
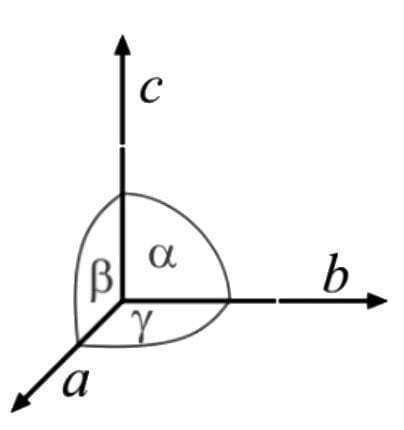
Reasoning (R): For a tetragonal system a = b = c and α = β = γ ≠ 90∘. The correct answer is
Reasoning (R): For a tetragonal system a = b = c and α = β = γ ≠ 90∘. The correct answer is
Detailed Solution: Question 7
How many number of atoms are there in a cube based unit cell having one atom on each corner and two atoms on each body diagonal of cube?
Detailed Solution: Question 8
The unit cell dimensions of a cubic lattice (edges a, b, c and the angles between them, α, β and γ) are
Detailed Solution: Question 9
A crystal made up of particles X, Y, and Z. X forms FCC& packing. Y occupies all octahedral voids of X and Z occupies all tetrahedral voids of X. If all particles along one body diagonal are removed, then the formula of the crystal is
Detailed Solution: Question 10
335 videos|697 docs|300 tests |



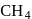 form molecular solid.
form molecular solid.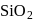 is a covalent network solid that forms a network of tetrahedral
is a covalent network solid that forms a network of tetrahedral 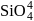 units.
units. 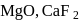 and
and 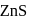 are ionic solids.
are ionic solids. atoms on the face-centres removing face-centred atoms along one of the
atoms on the face-centres removing face-centred atoms along one of the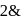 atoms.
atoms. atoms per unit cell
atoms per unit cell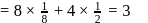
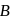 atoms per unit cell
atoms per unit cell 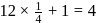
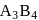
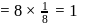
 & & & & Total number of atoms = 1 + 8 = 9.
& & & & Total number of atoms = 1 + 8 = 9.