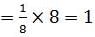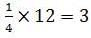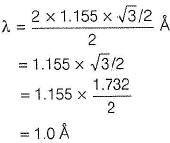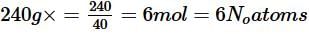Unit Cells, Lattices & Bragg's Equation (Old NCERT) - Free MCQ Practice
MCQ Practice Test & Solutions: Test: Unit Cells, Lattices & Bragg's Equation (Old NCERT) (20 Questions)
You can prepare effectively for JEE Chemistry for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Unit Cells, Lattices & Bragg's Equation (Old NCERT)". These 20 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 30 minutes
- - Number of Questions: 20
Sign up on EduRev for free to attempt this test and track your preparation progress.
Only One Option Correct Type
This section contains 9 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONLY ONE is correct.
Q. Bravais three-dimensional lattices are of
This section contains 9 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONLY ONE is correct.
Q. Bravais three-dimensional lattices are of
Detailed Solution: Question 1
The lattice site in a pure crystal cannot be occupied by
Detailed Solution: Question 2
Number of formula units in unit cells of MgO (rock salt), ZnS (zinc blende) and Pt (fee) respectively are
Detailed Solution: Question 3
A compound is formed by elements A and B and has cubic structure. A-atoms are at the corners and B-atoms are at the face centres. Thus, it is
Detailed Solution: Question 4
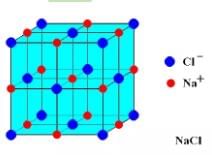
The mass of a unit cell of NaCL corresponds to
Detailed Solution: Question 5
At what angles for the first-order diffraction, spacing between two planes X respectively are λ and λ/2 ?
Detailed Solution: Question 6
A solid has a structure in which W-atoms are located at the corners of a cubic lattice, OF-atoms at the centre of edges and Na atom at the centre of the cube. The formula for the compound is
Detailed Solution: Question 7
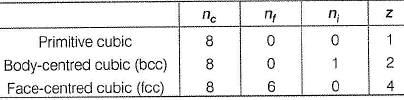
The number of atoms in primitive cubic unit cell, body-centred cubic unit cell and face-centred cubic unit cell are A , B and C respectively. Select the correct values.
Detailed Solution: Question 8
For a second order diffraction using X-ray B ragg’s equation, for a crystal with separation between two layers as d  , following graphical study was observed when varying 0 at different λ.
, following graphical study was observed when varying 0 at different λ.
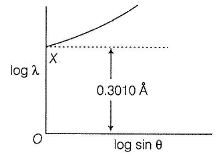
What is the value of d?
Detailed Solution: Question 9
One or More than One Options Correct Type
This section contains 3 multiple type questions. Each question has 4 choices (a), (b), (c) and (d), out of which ONE or MORE THAN ONE are correct.
In which of the following primitive cells axial distances or edge lengths (a, b, c)are different?
This section contains 3 multiple type questions. Each question has 4 choices (a), (b), (c) and (d), out of which ONE or MORE THAN ONE are correct.
In which of the following primitive cells axial distances or edge lengths (a, b, c)are different?
Detailed Solution: Question 10
Minimum interplanar spacing required for Bragg’s diffraction is:
Detailed Solution: Question 11
Which of the following Bravais lattices exist as face centered unit cell?
Detailed Solution: Question 12
Comprehension Type
This section contains a passage describing theory, experiments, data, etc. Two questions related to the paragraph have been given. Each question has only one correct answer out of the given 4 options (a), (b), (c) and (d).
Perovskite (A), a mineral containing calcium (o), oxygen  and titanium
and titanium  crystallises in the following cubic unit cell:
crystallises in the following cubic unit cell:
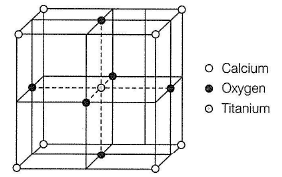
Q.
What is the Formula of A?
This section contains a passage describing theory, experiments, data, etc. Two questions related to the paragraph have been given. Each question has only one correct answer out of the given 4 options (a), (b), (c) and (d).
Perovskite (A), a mineral containing calcium (o), oxygen
Detailed Solution: Question 13
Perovskite (A), a mineral containing calcium (o), oxygen  and titanium
and titanium  crystallises in the following cubic unit cell:
crystallises in the following cubic unit cell:
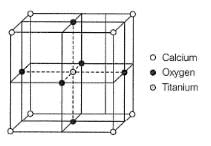
Q. What is the oxidation number of titanium in the given lattice?

Detailed Solution: Question 14
Matching List Type
Choices for the correct combination of elements from Column I and Column II are given as options (a), (b), (c) and (d), out of which one is correct.
Match the primitive unit cells given in Column I with their axial angles axial distances given in Column II and select the correct answer from the codes given
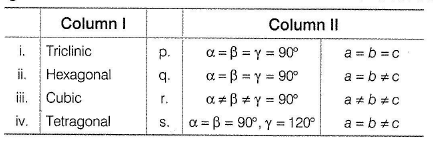

Choices for the correct combination of elements from Column I and Column II are given as options (a), (b), (c) and (d), out of which one is correct.
Detailed Solution: Question 15
One Integer Value Correct Type
This section contains 5 questions, when worked out will result in an integer value from 0 to 9 (both inclusive)
Q. A unit cell is characterised by how many parameters?
This section contains 5 questions, when worked out will result in an integer value from 0 to 9 (both inclusive)
Q. A unit cell is characterised by how many parameters?
Detailed Solution: Question 16
How many types of primitive unit cells are there?
Detailed Solution: Question 17
In which of the following Bravais lattices, not all axial angles are right angles?
Detailed Solution: Question 18
A second order Bragg’s diffraction of X-rays from a set of parallel planes separated by 1.155 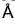 occurs at an angle 60° using wavelength of x
occurs at an angle 60° using wavelength of x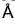 . What is the value of x?
. What is the value of x?
Detailed Solution: Question 19
Number of unit cells in 240 g of element X (atomic mass 40) which crystallises in bcc pattern in yN 0. What is the value of y?
Detailed Solution: Question 20
335 videos|697 docs|300 tests |