Werner's theory and Valence Bond - Free MCQ Practice Test with solutions,
MCQ Practice Test & Solutions: Test: Werner's theory and Valence Bond Theory (20 Questions)
You can prepare effectively for JEE Chemistry for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Werner's theory and Valence Bond Theory". These 20 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 20 minutes
- - Number of Questions: 20
Sign up on EduRev for free to attempt this test and track your preparation progress.
CN- is a strong field ligand. This is due to the fact that
Detailed Solution: Question 1
Which one of the following complexes is an outer orbital complex
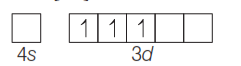
Atomic no.s Mn = 25, Fe = 26, Co = 27, Ni = 28
Atomic no.s Mn = 25, Fe = 26, Co = 27, Ni = 28
Detailed Solution: Question 2
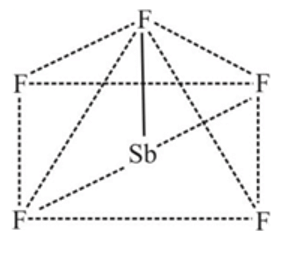
In the complex [SbF5]2- , sp3d hybridisation is present Geometry of the complex is
Detailed Solution: Question 3
An anion solution gives a white ppt with AgNO₃ solution. The ppt. dissolves in dil. ammonia due to the formation of
Detailed Solution: Question 4
Which of the following is incorrect regarding spectrochemical series?
Detailed Solution: Question 5
The spin only magnetic moment of [MnBr4]x− is 5.9BM. The geometry of the complex and x respectively are
Detailed Solution: Question 6
Which of the following are inner orbital complex (i.e., involving d2sp3 hybridisation) and is paramagnetic in nature?
Detailed Solution: Question 7
Which of the following is organo-metallic compound?
Detailed Solution: Question 8
The geometries of the ammonia complexes of Ni2+, Pt2+ and Zn2+, respectively, are
Detailed Solution: Question 9
[NiCl2{P(C2H5)2(C6H5)}2] exhibits temperature dependent magnetic behaviour (paramagnetic/diamagnetic). The coordination geometries of Ni2+ in the paramagnetic and diamagnetic states are respectively
Detailed Solution: Question 10
Which of the following complex compound(s) is/are paramagnetic and low spin?
(I) K₃[Fe(CN)₆]
(II) [Ni(CO)₄]⁰
(III) [Cr(NH₃)₆]³⁺
(IV) [Mn(CN)₆]⁴⁻
Choose the correct code:
(I) K₃[Fe(CN)₆]
(II) [Ni(CO)₄]⁰
(III) [Cr(NH₃)₆]³⁺
(IV) [Mn(CN)₆]⁴⁻
Choose the correct code:
Detailed Solution: Question 11
An aqueous solution of titanium chloride, when subjected to magnetic measurement, measured zero magnetic moment. Assuming the octahedral complex in aqueous solution, the formulae of the complex is:
Detailed Solution: Question 12
Which of the following statement is not true for the reaction given below?
[Cu(H2O)4]²⁺ + 4NH3 ⇌ [Cu(NH3)4]²⁺ + 4H2O
[Cu(H2O)4]²⁺ + 4NH3 ⇌ [Cu(NH3)4]²⁺ + 4H2O
Detailed Solution: Question 13
Atomic number of Cr, Fe and Co are 24, 26 and 27 respectively. Which of the following inner orbital octahedral complexes are paramagnetic?
Detailed Solution: Question 14
Detailed Solution: Question 15
Which of the following statements is incorrect?
Detailed Solution: Question 16
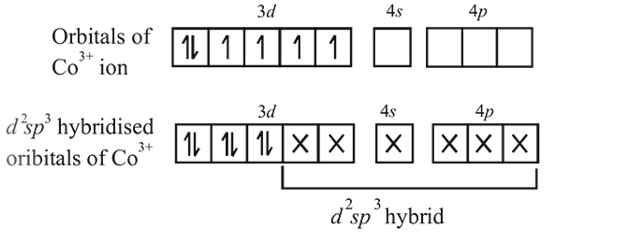
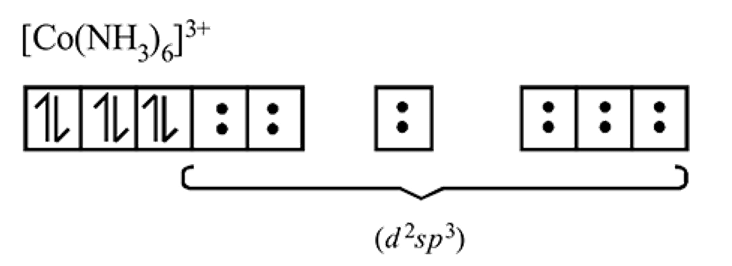
An octahedral complex of CO3+ is diamagnetic. The hybridisation involved in the formation of the complex is:
Detailed Solution: Question 17
The correct statement about the magnetic properties of [Fe(CN)₆]³⁻ and [FeF₆]³⁻ is:
(Z = 26):
(Z = 26):
Detailed Solution: Question 18
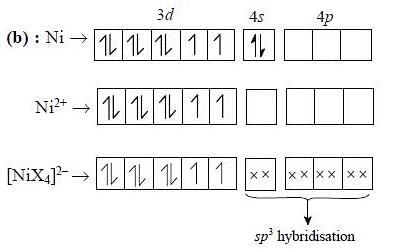
Nickel (Z = 28) combines with a uninegative monodentate ligand to form a diamagnetic complex [NiX4]2-. The hybridization involved and the number of unpaired electrons present in the complex is respectively:
Detailed Solution: Question 19
Prussian blue is a deep blue pigment containing Fe2+, Fe3+ and CN- ions. It has the formula Fe7(CN)18. How many Fe2+ and Fe3+ ions are there per formula unit?
Detailed Solution: Question 20
332 videos|699 docs|300 tests |


 bond from pseudohalide to the metal and
bond from pseudohalide to the metal and  bond (from the metal to pseudohalide).
bond (from the metal to pseudohalide).
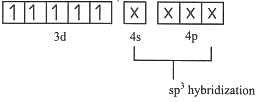
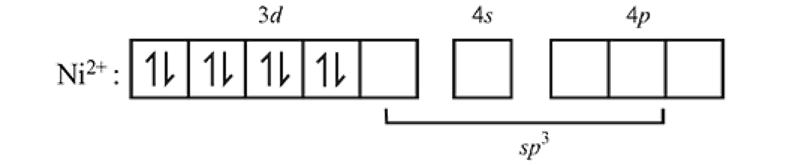
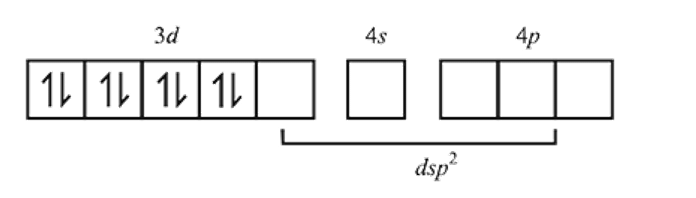
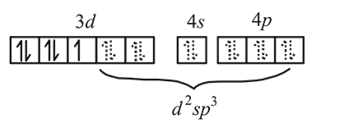
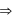 low spin complex μ = 1.732BM Ni in [Ni(CO)4]
low spin complex μ = 1.732BM Ni in [Ni(CO)4]
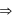 low and high spin complex is applicable for d4 to d7 configuration
low and high spin complex is applicable for d4 to d7 configuration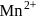 in
in 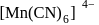

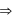 low spin complex μ = 1.732BM
low spin complex μ = 1.732BM