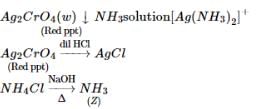d & f-Block Elements - 2 - Free MCQ Practice Test with solutions, NEET
MCQ Practice Test & Solutions: Test: d & f-Block Elements - 2 (30 Questions)
You can prepare effectively for NEET NCERT Based Tests for NEET with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: d & f-Block Elements - 2". These 30 questions have been designed by the experts with the latest curriculum of NEET 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 45 minutes
- - Number of Questions: 30
Sign up on EduRev for free to attempt this test and track your preparation progress.
(T) imparts violet colour 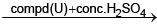 (V) Red gas
(V) Red gas 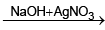 (W) Red ppt.
(W) Red ppt.
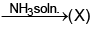
(W) Red ppt. 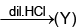 white ppt.
white ppt.
(U) 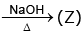 gas (gives white fumes with HCl)
gas (gives white fumes with HCl)
sublimes on heating Identify (T) to (Z).
Detailed Solution: Question 1
The number of moles of acidified KMnO4 required to convert one mole of sulphite ion into sulphate ion is-
Detailed Solution: Question 2
N2(g) + 3H2 (g) 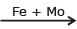 2NH3(g) ; Haber's process, Mo is used as -
2NH3(g) ; Haber's process, Mo is used as -
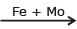 2NH3(g) ; Haber's process, Mo is used as -
2NH3(g) ; Haber's process, Mo is used as -Detailed Solution: Question 3
Potash alum is a double salt, its aqueous solution shows the characteristics of-
Detailed Solution: Question 4
Detailed Solution: Question 5
Addition of non-metals like B and C to the interstitial sites of a transition metal results the metal-
Detailed Solution: Question 6
Detailed Solution: Question 7
Detailed Solution: Question 8
The correct statement(s) about transition elements is/are-
Detailed Solution: Question 9
An ornament of gold having 75% of gold, it is of .............. carat.
Detailed Solution: Question 10
Detailed Solution: Question 11
The ionisation energies of transition elements are-
Detailed Solution: Question 12
Transition elements having more tendency to form complex than representative elements (s and p-block elements) due to-
Detailed Solution: Question 13
During estimation of oxalic acid Vs KMnO4, self indicator is-
Detailed Solution: Question 14
The metal(s) which does/do not form amalgam is/are-
Detailed Solution: Question 15
Which of the following statements concern with transition metals ?
Detailed Solution: Question 16
Detailed Solution: Question 17
The highest oxidation state among transition elements is-
Detailed Solution: Question 18
A compound of mercury used in cosmetics, in Ayurvedic and Yunani medicines and known as Vermilon is-
Detailed Solution: Question 19
Acidified chromic acid 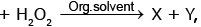
X and Y are - (blue colour)
Detailed Solution: Question 20
Detailed Solution: Question 21
Transition elements are usually characterised by variable oxidation states but Zn does not show this property because of-
Detailed Solution: Question 22
(NH4)2Cr2O7 (Ammonium dichromate) is used in fire works. The green coloured powder blown in air is-
Detailed Solution: Question 23
The d-block element which is a liquid at room temperature, having high specific heat, less reactivity than hydrogen and its chloride (MX2) is volatile on heating is-
Detailed Solution: Question 24
Detailed Solution: Question 25
Iron becomes passive by..................due to formation of ................
Detailed Solution: Question 26
Bayer's reagent used to detect olifinic double bond is-
Detailed Solution: Question 27
Detailed Solution: Question 28
Detailed Solution: Question 29
Detailed Solution: Question 30