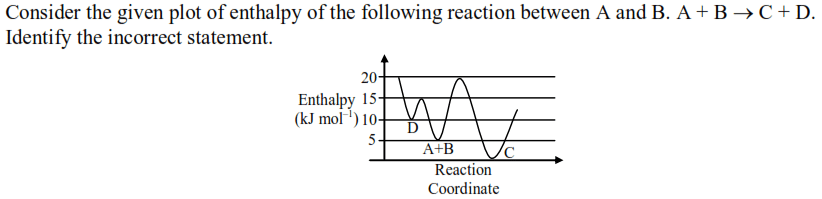
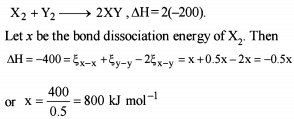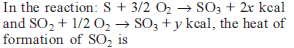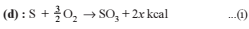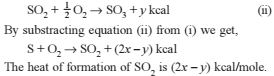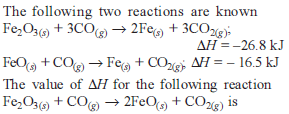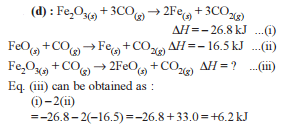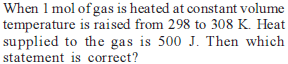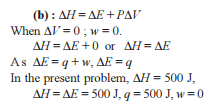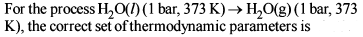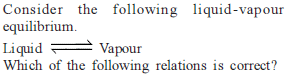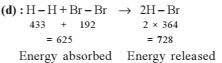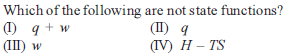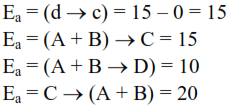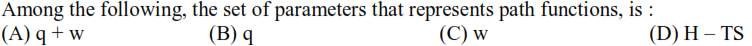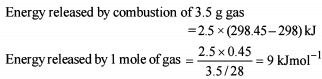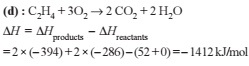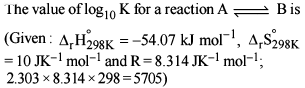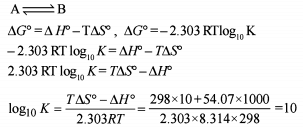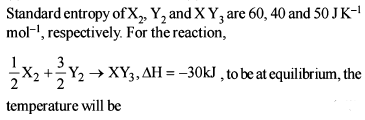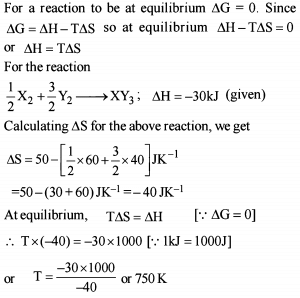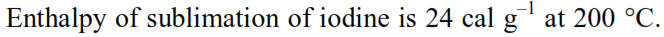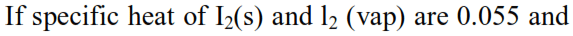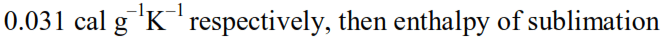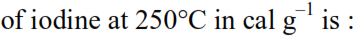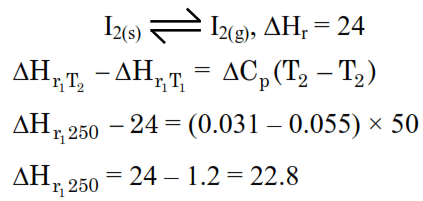Thermodynamics - 2 - Free MCQ Test with solutions for JEE Chemistry
MCQ Practice Test & Solutions: Thermodynamics - 2 (30 Questions)
You can prepare effectively for JEE Chemistry for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Thermodynamics - 2". These 30 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 60 minutes
- - Number of Questions: 30
Sign up on EduRev for free to attempt this test and track your preparation progress.
3 moles of a diatomic gas are heated from 127° C to 727° C at a constant pressure of 1 atm. Entropy change is (log 2.5 = 0 .4)
Detailed Solution: Question 1
Exactly 100 J of heat was transferred reversibly to a block of gold at 25.00° C from a thermal reservoir at 25.01 °C, and then exactly 100 J of heat was absorbed reversibled from the block of gold by a thermal reservoir at 24.99° C. Thus entropy change of the system is
Detailed Solution: Question 2
Consider a reversible isentropic expansion of 1.0 mole of an ideal monoatomic gas from 25°C to 75°C. If the initial pressure was 1.0 bar, final pressure is
Detailed Solution: Question 3
For the process,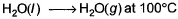 and 1 atmosphere pressure, the correct choice is
and 1 atmosphere pressure, the correct choice is
[JEE Advanced 2014]
Detailed Solution: Question 4
Benzene and naphthalene form an ideal solution at room temperature. For this process, the true statement(s) is (are)
[JEE Advanced 2013]
Detailed Solution: Question 5
Detailed Solution: Question 6
Detailed Solution: Question 7
Detailed Solution: Question 8
Detailed Solution: Question 9
Detailed Solution: Question 10
Detailed Solution: Question 11
Detailed Solution: Question 12
Detailed Solution: Question 13
Detailed Solution: Question 14
Detailed Solution: Question 15
Detailed Solution: Question 16
Detailed Solution: Question 17
Detailed Solution: Question 18
Detailed Solution: Question 19
Detailed Solution: Question 20
Detailed Solution: Question 21
Detailed Solution: Question 22
Which one of the following equations does not correctly represent the first law of thermodynamics for the given processes involving an ideal gas ? (Assume non-expansion work is zero)
Detailed Solution: Question 23
Detailed Solution: Question 24
Detailed Solution: Question 25
Detailed Solution: Question 26
Detailed Solution: Question 27
Detailed Solution: Question 28
Detailed Solution: Question 29
Detailed Solution: Question 30
335 videos|699 docs|300 tests |