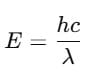WBJEE Sample Papers, Section Wise & Full Chemistry Test - 2 Free Online
MCQ Practice Test & Solutions: WBJEE Chemistry Test - 2 (40 Questions)
You can prepare effectively for JEE WBJEE Sample Papers, Section Wise & Full Mock Tests 2026 with this dedicated MCQ Practice Test (available with solutions) on the important topic of "WBJEE Chemistry Test - 2". These 40 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 60 minutes
- - Number of Questions: 40
Sign up on EduRev for free to attempt this test and track your preparation progress.
Which of the following species can act both as Bronsted acid and base ? NH₂⁻ 2. HCO₃⁻ 3. NH₄⁺ 4. HSO₄⁻ Select the correct answer using the codes given below :
Detailed Solution: Question 1
Detailed Solution: Question 3
Which of the following is not a preliminary test used to detect ions
The wavelength of light waves is inversely proportional to its
Detailed Solution: Question 6
Which of the following alkyl group has the maximum + I effect?
The enzymes which convert glucose and fructose into ethyl alcohol is
The enthalpies of combustion of carbon and carbon monoxide are -393.5 and -283 KJ mol⁻¹ respectively .The enthalpy of formation of carbon monoxide per mol is
A glass bulb is filled with NO₂ gas and immersed in an ice bath at 0°C which becomes colourless after some time . This colourless gas will be
Units of rate constant for zero- and first order reaction in terms of molarity (M) units are, respectively,
Detailed Solution: Question 12
The soaps are salts of higher fatty acids containing chains of
Detailed Solution: Question 16
The process associated with manufacture of sodium carbonate is known as
The density of gas at 27°C and 1 atm is d. Pressure remaining constant at which of the following temperatures will its density become 0.75 d ?
Detailed Solution: Question 18
What is formed when boron is heated with concentrated H₂SO₄?
Which one of the following substances is used in the laboratory for a fast drying of neutral gases?
Which of the following isomerism is not exhibited by alkanes?
Detailed Solution: Question 24
The crystal system of a compound with unit cell dimensions a=0.387, b=0.387 and c=0.0504 nm and α = β = 90°and γ = 120°is
In the Fischer projection formula, the representation of meso-butane-2,3-diol is :
3 videos|21 docs|54 tests |
3 videos|21 docs|54 tests |