WBJEE Sample Papers, Section Wise & Full Chemistry Test - 6 Free Online
MCQ Practice Test & Solutions: WBJEE Chemistry Test - 6 (40 Questions)
You can prepare effectively for JEE WBJEE Sample Papers, Section Wise & Full Mock Tests 2026 with this dedicated MCQ Practice Test (available with solutions) on the important topic of "WBJEE Chemistry Test - 6". These 40 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 60 minutes
- - Number of Questions: 40
Sign up on EduRev for free to attempt this test and track your preparation progress.
Detailed Solution: Question 1
Which of the following does not turn Schiff's reagent to pink ?
Sometimes yellow turbidity appears while passing H₂S gas even in the absence of II group radicals. This is because of
The energy of second Bohr orbit of the hydrogen atom is -328KJ mol⁻1. The energy of fourth Bohr orbit would be
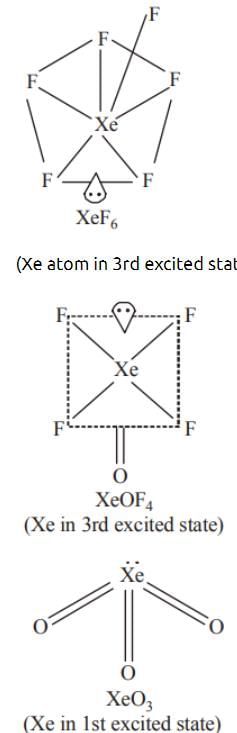
Among the molecules (a)XeO₃ (b)XeOF (c)XeOF₆,those having same no. of lone pairs on Xe are
Detailed Solution: Question 8
The heat of formation of CO₂ is -94 Kcal mol⁻1 whereas ∆H for 2CeO₂ → 2CO is -110.5 Kcal,The enthalpy of combustion carbon is
The equilibrium constant for the following reaction will be 3 A + 2 B → C
Detailed Solution: Question 11
A reaction with respect to the reactant A has a rate constant of 5 s⁻1. If we start with[A]= 10 mol-L⁻1, then in what time the concentration of A becomes 1.0 mol-L⁻1?
In an electrochemical cell, if Eº is the EMF of the cell involving 'n' mole of electrons, then ∆Gº is
The number and type of bonds between two carbon atoms in calcium carbide are
Electromagnetic separation is used in the concentration of
A real gas most closely approaches the behaviour of an ideal gas at
What are the products obtained when ammonia is treated with excess of chlorine ?
Sodium sulphate is soluble in water but barium sulphate is insoluble because
The isomers, which possess same structural formula, but differ in spatial arrangement of the group around the double bond, are called
The radioisotope, tritium (3H₁) has a half-life of 12.3 years. If the initial amount of tritium is 32 mg, how many miligrams of it would remain after 49.2 years ?
In which of the following reaction there is no change in oxidation number
Consider the following statements about enantiomers :
1. Isomers which are mirror images of each other are called enantiomers
2. A mixture of equal parts of enantiomers is called a racemic modification
3. Enantiomers show different properties-physical or chemical -only in a chiral medium
4. A racemic modification is converted by an optically active agent into mixture of diasteromers that can be separated.
Which of the above statements are correct ?
3 videos|21 docs|54 tests |
3 videos|21 docs|54 tests |