Thermodynamics of Electrochemistry - Free MCQ Practice Test with solutions,
MCQ Practice Test & Solutions: Test: Thermodynamics of Electrochemistry (24 Questions)
You can prepare effectively for NEET NCERT Based Tests for NEET with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Thermodynamics of Electrochemistry". These 24 questions have been designed by the experts with the latest curriculum of NEET 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 35 minutes
- - Number of Questions: 24
Sign up on EduRev for free to attempt this test and track your preparation progress.
Only One Option Correct Type
This section contains 15 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONLY ONE is correct
Q.
Temperature coefficient of EMF of a cell in terms of entropy change is
Detailed Solution: Question 1
The standard reduction potential at 298 K of the reaction,
2H2O + 2e-  H2 + 2OH- is 0.8277 V
H2 + 2OH- is 0.8277 V
Thus,thermodynamic equilibrium constant for the reaction.
2H2O  H3O+ + OH- at 298 K is
H3O+ + OH- at 298 K is
 H2 + 2OH- is 0.8277 V
H2 + 2OH- is 0.8277 V H3O+ + OH- at 298 K is
H3O+ + OH- at 298 K is Detailed Solution: Question 2
Given ,
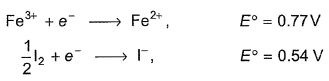
Thus ,equilibrium constant for the reaction in terms of log k is
2Fe3+ + 3I-  2Fe2+ + I-3
2Fe2+ + I-3
 2Fe2+ + I-3
2Fe2+ + I-3 Detailed Solution: Question 3
Given ,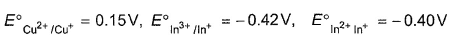
Thus ,(log Keq) for the reaction Cu2+ +In2+  Cu+ + In3+ is
Cu+ + In3+ is
 Cu+ + In3+ is
Cu+ + In3+ is Detailed Solution: Question 4
An excess of liquid mercury is added to an acidified solution of 1.0 x 10-3 M Fe3+ .Thus 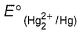 is if 5% of Fe3+ remains at equilibrium at 298 K
is if 5% of Fe3+ remains at equilibrium at 298 K
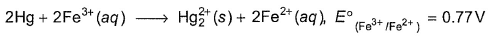
Detailed Solution: Question 5
EMF of the following cell is 0.2905 V
Zn/Zn2+ (a = 0.1M)|| Fe2+ (a = 0.01M)| Fe
The equilibrium constant for the cell reaction is
[IIT JEE 2004]
Detailed Solution: Question 6
The half-cell reactions for rusting of iron are
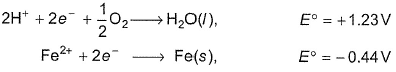
ΔG° (in kJ) for the reaction is
[IIT JEE 2005]
Detailed Solution: Question 7
The Gibbs free energy for the decomposition of Al2O3 at 800 K is as follows :
2Al2O3 → 4Al + 3O2 , ΔrG = 2898kJ mol-1
The potential difference needed for electrolytic reduction of Al2O3 is at least
Detailed Solution: Question 8
Given , 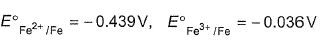
The value of standard electrode potential for the half-reaction is
Fe3+(aq) + e- → Fe2+(aq)
Detailed Solution: Question 9
For the reaction, 2H2(g) + O2(g) → 2H2O(l), E0cell = 1.23 V at 298 K
and ΔH0f (H2O) = - 285.8 kJ mol-1 Thus, ΔS° (standard entropy change ) is
Detailed Solution: Question 10
For the reaction ,
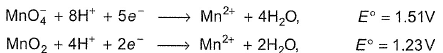
Thus , for the reaction
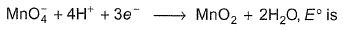
Detailed Solution: Question 11
Consider the following equations for a cell reaction.
A+B  C+ D, E0 = x volt, Keq = K1
C+ D, E0 = x volt, Keq = K1
2A +2B  2C+ 2D, E0 = y volt, Keq = K2
2C+ 2D, E0 = y volt, Keq = K2
Then,
 C+ D, E0 = x volt, Keq = K1
C+ D, E0 = x volt, Keq = K1 2C+ 2D, E0 = y volt, Keq = K2
2C+ 2D, E0 = y volt, Keq = K2Detailed Solution: Question 12
Which of the following statements about the spontaneous reaction occurring in a galvanic cell is always true?
Detailed Solution: Question 13
For a (Ag-Zn) button cell ,the net reaction is
Zn(s) + Ag2O(s) → ZnO(s) + 2Ag(s)
ΔG0f(Ag2O) = -11.21kJmol-1, ΔG0 f(ZnO) = - 318.3 kJ mol-1
Hence, E°cell of the button cell is
Detailed Solution: Question 14
Detailed Solution: Question 15
Matching List Type
Choices for the correct combination of elements from Column I and Column II are given as options (a), (b), (c) and (d), out of which one is correct
Q.
The standard reduction potential data at 298 K is given below:
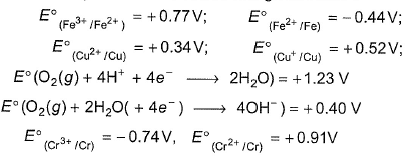
Match E° of a redox pair in Column I with the values given in Column II and select the corect answer using the codes given below:


Detailed Solution: Question 16
The standard potential of the following cell is 0.23 V at 288 K and 0.21 V at 308 K.
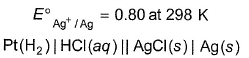
Match the parameters in Column I with their values in Column II and select the answer from the codes given below the list.
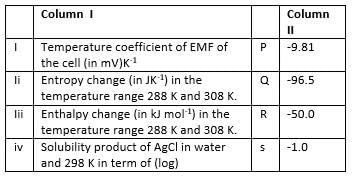


Detailed Solution: Question 17
Comprehension Type
This section contains a passage describing theory, experiments, data, etc. Two questions related to the paragraph have been given. Each question has only one correct answer out of the given 4 options (a), (b), (c) and (d)
Passage I
The concentration of potassium ions inside a biological cell is at least twenty times higher than the outside. The resulting potential difference across the cell is important in several process such as transmission of nerve impulses and maintaining the ion balance. A simple model for such a concentration cell involving a metal M is
M(s) | M+ (aq, 0.05M), || M+(aq) 1M|M(s) | Ecell | = 77mV
Q.
For the above cell,
Detailed Solution: Question 18
Passage I
The concentration of potassium ions inside a biological cell is at least twenty times higher than the outside. The resulting potential difference across the cell is important in several process such as transmission of nerve impulses and maintaining the ion balance. A simple model for such a concentration cell involving a metal M is
M(s) | M+ (aq, 0.05M), || M+(aq) 1M|M(s) | Ecell | = 77mV
Q.
If 0.05 M solution of M+ is replaced by 0.0025 M solution of M+,then |Ecell | would be
Detailed Solution: Question 19
Passage II
Given,
ΔG0f(AgCl) = -109kJmol-1, ΔG0f(Cl-) = -129kJmol-1
ΔG0f(Ag+) = 77kJmol-1,
Thus E°cell of the cell reaction is
Ag+(aq) + Cl-(aq) → AgCl(s) is
Detailed Solution: Question 20
Passage II
Given,
ΔG0f(AgCl) = -109kJmol-1, ΔG0f(Cl-) = -129kJmol-1
ΔG0f(Ag+) = 77kJmol-1,
Q.
Ksp of AgCl is thus,
Detailed Solution: Question 21
One Integer Value Correct Type
This section contains 3 questions, when worked out will result in an integer value from 0 to 9 (both inclusive)
Q.
A platinum electrode is immersed in a solution containing 0.1 M Fe2+ and 0.1 M Fe3+.It iscoupled with SHE.Concentration Fe3+ of increased to 0.1 M without change in [Fe2+], then the change in EMF (in centivolt) is
Detailed Solution: Question 22
Using Cr2O72- aqueous, solution
E0red = 1.33V and ΔG0 = -770.07 kJmol-1
What is the valency of the ion formed after reduction?
E0red = 1.33V and ΔG0 = -770.07 kJmol-1
Detailed Solution: Question 23
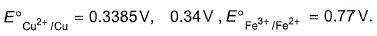
Equilibriumconstant of the cell reaction,
Cu + 2Fe3+  2Fe2+ + cu2+ is y x 1014
2Fe2+ + cu2+ is y x 1014
What is the value of y?
 2Fe2+ + cu2+ is y x 1014
2Fe2+ + cu2+ is y x 1014Detailed Solution: Question 24



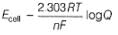
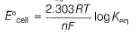
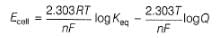
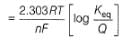


 Ag+ + Cl- E0 = -0.59V
Ag+ + Cl- E0 = -0.59V