Addition & Condensation Polymers (Old NCERT) - Free MCQ Practice Test
MCQ Practice Test & Solutions: Test: Addition & Condensation Polymers (Old NCERT) (10 Questions)
You can prepare effectively for JEE Chemistry for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Addition & Condensation Polymers (Old NCERT)". These 10 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 15 minutes
- - Number of Questions: 10
Sign up on EduRev for free to attempt this test and track your preparation progress.
Detailed Solution: Question 1
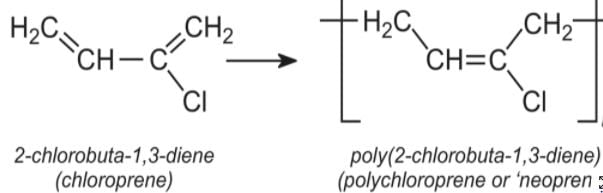
A synthetic polymer which resembles natural rubber is:
Detailed Solution: Question 2
Which type of polymer will be formed if the susbstituent group is –C6H5
Detailed Solution: Question 3
Vulcanisation is used in processing of:
Detailed Solution: Question 4
Detailed Solution: Question 5
Detailed Solution: Question 6
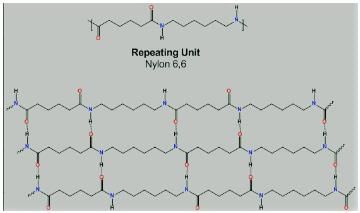
Which intermolecular force is present in Nylon 6,6?
Detailed Solution: Question 7
Polymers having ester linkage are called:
Detailed Solution: Question 8
Detailed Solution: Question 9
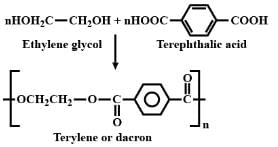
Condensation polymerisation of ethylene glycol and terepthalic acid gives:
Detailed Solution: Question 10
335 videos|699 docs|300 tests |