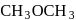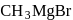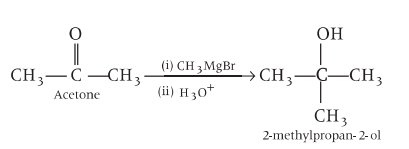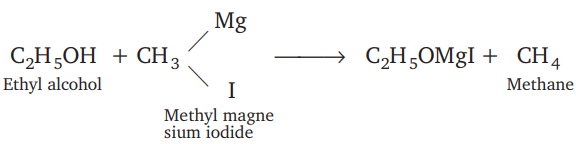Preparation and Properties of Alcohols - Free MCQ Practice Test with solutions,
MCQ Practice Test & Solutions: Test: Preparation and Properties of Alcohols (20 Questions)
You can prepare effectively for JEE Chemistry for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Preparation and Properties of Alcohols". These 20 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 20 minutes
- - Number of Questions: 20
Sign up on EduRev for free to attempt this test and track your preparation progress.
How many structural isomers of 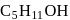 will be primary alcohols?
will be primary alcohols?
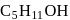 will be primary alcohols?
will be primary alcohols?Detailed Solution: Question 1
Which of the following species can act as the strongest base?
Detailed Solution: Question 2
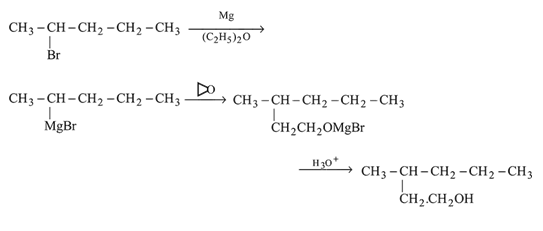
Which of the following synthesis gives 3 -methyl-1- hexanol?
Detailed Solution: Question 3
Detailed Solution: Question 4
Which of the following are intermediates in the reaction of excess of 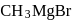 with
with 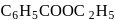 to make 2 -phenyl
to make 2 -phenyl 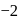 -propanol?
-propanol?
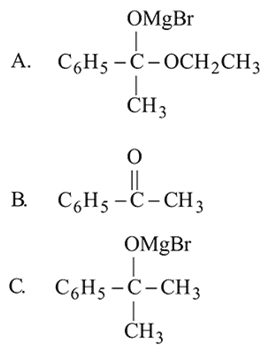
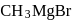 with
with 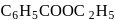 to make 2 -phenyl
to make 2 -phenyl 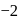 -propanol?
-propanol?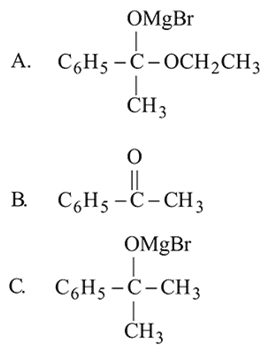
Detailed Solution: Question 5
Detailed Solution: Question 6
Which one/ones of the following reactions will yield 2-propanol?
I. 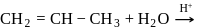
II.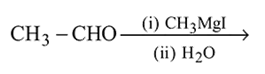
III.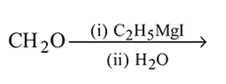
IV. 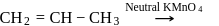
I.
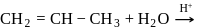
II.
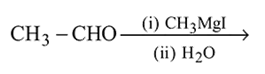
III.
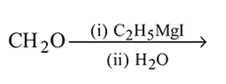
IV.
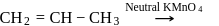
Detailed Solution: Question 7
Which of the following has lowest boiling point?
Detailed Solution: Question 8
The following change can be carried out with
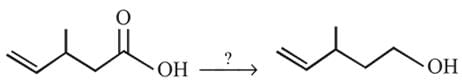
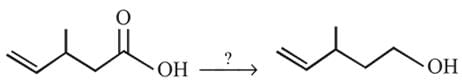
Detailed Solution: Question 9
Detailed Solution: Question 10
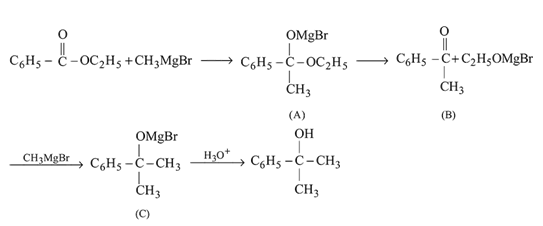
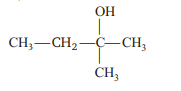
Ethyl magnesium bromide reacts with acetone to give X. On hydrolysis X forms
Detailed Solution: Question 11
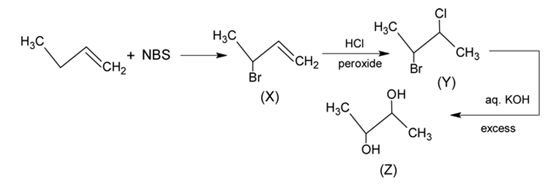
Identify the product [Z] in the given sequence of reaction
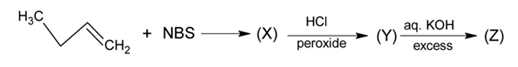
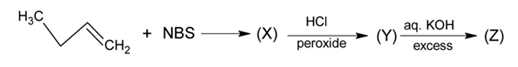
Detailed Solution: Question 12

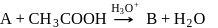
In the above reactions ' 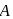 ' and '
' and ' 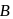 ' respectively are
' respectively are

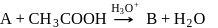
In the above reactions '
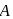 ' and '
' and ' 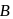 ' respectively are
' respectively areDetailed Solution: Question 13
Detailed Solution: Question 14
The boiling points of isomeric alcohols follow the order
Detailed Solution: Question 15
Detailed Solution: Question 16
Detailed Solution: Question 17
Identify the suitable reagent for the reaction given below.
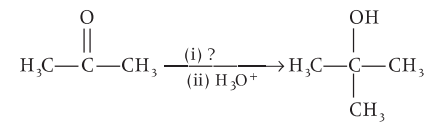
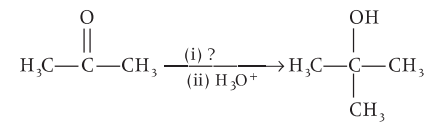
Detailed Solution: Question 18
Detailed Solution: Question 19
Which one of the following gases is liberated when ethyl alcohol is heated with methyl magnesium iodide?
Detailed Solution: Question 20
335 videos|699 docs|300 tests |


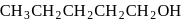
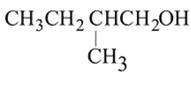
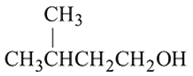
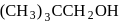

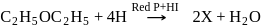 ;
;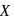 is
is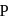 and HI to alkanes through alkyl iodides
and HI to alkanes through alkyl iodides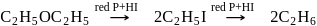
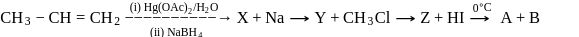
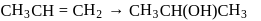
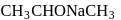
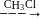
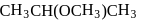
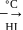

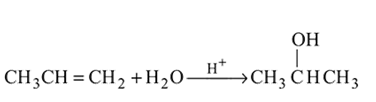
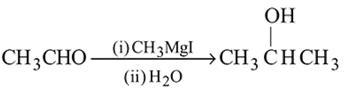
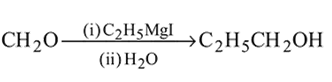
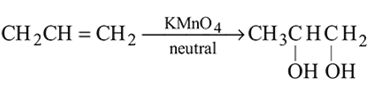
 -Nitrophenol has intramolecular H-bonding.
-Nitrophenol has intramolecular H-bonding.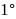 alcohols using Lithium aluminum hydride
alcohols using Lithium aluminum hydride 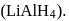 An aldehyde is produced as an intermediate during this reaction, but it cannot be isolated because it is more reactive than the original carboxylic acid.
An aldehyde is produced as an intermediate during this reaction, but it cannot be isolated because it is more reactive than the original carboxylic acid.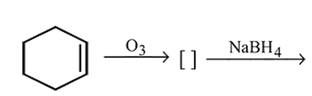
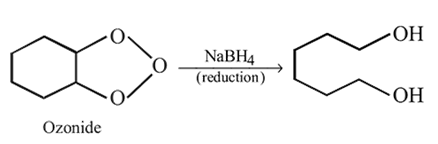
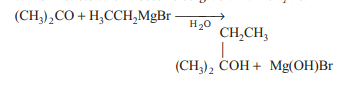
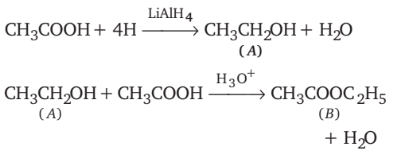
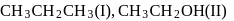 and
and 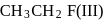 is in order
is in order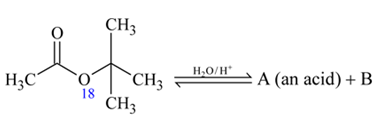
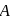 and
and  are, respectively,
are, respectively,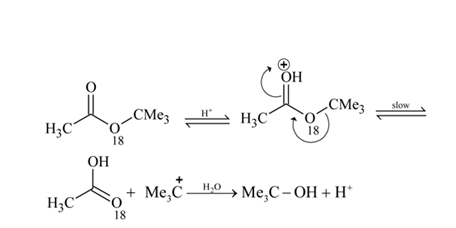
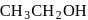 and
and