JEE Main Chemistry Test- 3 Free Online Test 2026
MCQ Practice Test & Solutions: JEE Main Chemistry Test- 3 (25 Questions)
You can prepare effectively for JEE Mock Tests for JEE Main and Advanced 2026 with this dedicated MCQ Practice Test (available with solutions) on the important topic of "JEE Main Chemistry Test- 3". These 25 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 60 minutes
- - Number of Questions: 25
Sign up on EduRev for free to attempt this test and track your preparation progress.
Detailed Solution: Question 1
The nature of inter-molecular forces among benzene molecules is
Detailed Solution: Question 3
The addition of HCN to a carbonyl compound is an example of
Detailed Solution: Question 5
In the presence of platinum catalyst, hydrocarbon A adds hydrogen to form n-hexane. When hydrogen bromide is added to A instead of hydrogen, only a single bromo compound is formed. Which of the following is A ?
The alkene 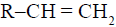 reacts readily with
reacts readily with 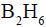 and formed the product B which on oxidation with alkaline hydrogen peroxides produces
and formed the product B which on oxidation with alkaline hydrogen peroxides produces
Amongst the following, the compound that can be most readily sulphonated is
Detailed Solution: Question 12
Detailed Solution: Question 14
Which of the following compound will make precipitate most readily with 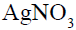 ?
?
Which of the following is responsible for depletion of the ozone layer in the upper strata of the atmosphere?
7.5 grams of a gas occupy 5.6 litres of volume at STP. The gas is
Detailed Solution: Question 17
The number of water molecules present in a drop of water (volume 0.0018 ml) at room temperature is
Detailed Solution: Question 18
The correct representation of Charle’s law is given by
The structure of TlCl is similar to CsCl. What would be the radius ratio in TlCl ?
Sulphur trioxide is prepared by the following two reactions:-
S8(s) + 8 O2(g) —→ 8 SO2(g)
2SO2(g) + O2(g) —→ 2 SO3(g)
How many grams of SO3 are produced from 1 mole S8?
S8(s) + 8 O2(g) —→ 8 SO2(g)
2SO2(g) + O2(g) —→ 2 SO3(g)
How many grams of SO3 are produced from 1 mole S8?
Detailed Solution: Question 21
How many elements are more electropositive than Cl?
Be, F, O, S, P, Au, H, Na
Be, F, O, S, P, Au, H, Na
Detailed Solution: Question 22
How many elements have more ionisation energy as compared to their next higher atomic number element?
Na, Mg, Al, Si, P, S, Cl, Ar
Na, Mg, Al, Si, P, S, Cl, Ar
Detailed Solution: Question 23
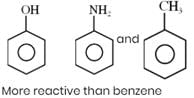
How many of the following compounds are more reactive than benzene towards electrophilic substitution.
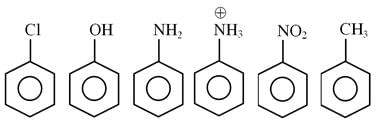

Detailed Solution: Question 24
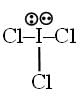
Number of d-orbitals used in the hybridisation of ICl3 is = x and number of lone pair at central atom = y find x + y = ?
Detailed Solution: Question 25
360 docs|100 tests |