JEE Main & Advanced Chemistry Test- 12 Free Online Test 2026
MCQ Practice Test & Solutions: JEE Main Chemistry Test- 12 (30 Questions)
You can prepare effectively for JEE JEE Main & Advanced Mock Test Series 2026 with this dedicated MCQ Practice Test (available with solutions) on the important topic of "JEE Main Chemistry Test- 12". These 30 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 60 minutes
- - Number of Questions: 30
Sign up on EduRev for free to attempt this test and track your preparation progress.
Detailed Solution: Question 1
10 mL of gaseous hydrocarbon on combustion gives 40 mL of 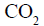 (g) and 50 mL of
(g) and 50 mL of 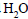 (vap.).The
(vap.).The
hydrocarbon is
0.7 g of 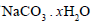 is dissolved in 10 mL of water, 20mL of which required 19.8 mL of 0.1 N HCl. The value of x is
is dissolved in 10 mL of water, 20mL of which required 19.8 mL of 0.1 N HCl. The value of x is
The energy of the second Bohr orbit of the hydrogen atom is –3.41 eV. The energy of the third Bohr orbit of the  ion will be
ion will be
The energy of an electron in the first Bohr orbit of H-atom is –13.6 eV. The possible energy values(s)
of the excited state(s) for electrons in Bohr orbits of hydrogen is (are)
of the excited state(s) for electrons in Bohr orbits of hydrogen is (are)
The ratio of time periods in first and second orbits of hydrogen atom is
The sub-shell that comes after f-sub-shell is called g-sub-shell. The number of g-sub orbitals in g-sub shell and the total number of orbitals in the principal orbital respectively are
The e/m for positive rays in comparison to cathod rays is
If n and l are respectively the principal and azimuthal quantum numbers, then the expression for calculating total number of electrons in any energy level is
Detailed Solution: Question 13
Name the two elements that are liquid at ordinary temperature
Traid-I [N3-, Na+]
Traid-II [N+, C+,O+] Choose the species of lowers IP from traid-I and the species of highest IP from traid-II respectively
Traid-II [N+, C+,O+] Choose the species of lowers IP from traid-I and the species of highest IP from traid-II respectively
Detailed Solution: Question 17
Arrange the elements with the following electronic configurations in increasing order of electron affinity 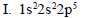
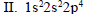
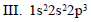
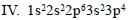
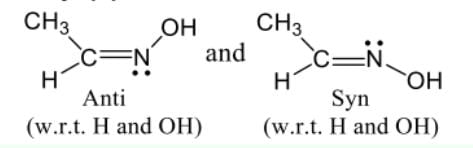
Which of the following is the correct order of stability of the following four distinct conformations of nbutane?
Which among the following is likely to show geometrical isomerism?
Detailed Solution: Question 24
Which one of the following is the stablest structure of cyclohexatriene ?
meso-Tartaric acid is optically inactive due to the presence of
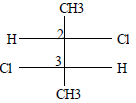
The absolute cofiguration of the following compound is
1 videos|238 docs|217 tests |