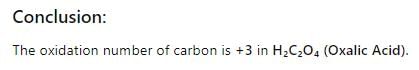JEE Main & Advanced Chemistry Test- 6 Free Online Test 2026
MCQ Practice Test & Solutions: JEE Main Chemistry Test- 6 (25 Questions)
You can prepare effectively for JEE JEE Main & Advanced Mock Test Series 2026 with this dedicated MCQ Practice Test (available with solutions) on the important topic of "JEE Main Chemistry Test- 6". These 25 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 60 minutes
- - Number of Questions: 25
Sign up on EduRev for free to attempt this test and track your preparation progress.
System which suggested Roman numbers to name compounds is called as
Detailed Solution: Question 1
Detailed Solution: Question 2
Suppose a compound contains atoms A,B and C. The
oxidation number of A is 2, B is 5 and C is –2.
The possible formula of the compound would be
Detailed Solution: Question 3
Which of the following ions will undergo
disproportionation reaction ?
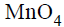 is good oxidizing agent in different media,where it changes to
is good oxidizing agent in different media,where it changes to

The oxidatino number of Mn correspondingly reduces by
Detailed Solution: Question 11
The hyridization states of the central atom in the complex
ions 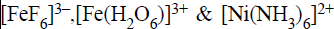 are
are
Consider the following spatial arrangements of the
octahedral complex ion 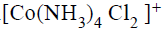
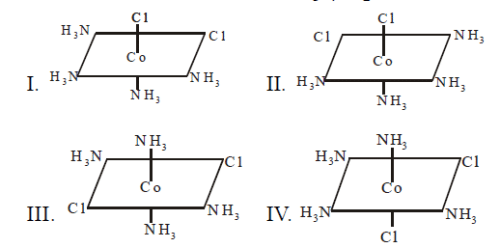
Which of the following statement is incorrect
regarding these stuructures ?
The chloride ion is detected by the formation of a white
precipitate of AgCl on addition of aqueous 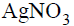
solution and by the dissociation of the precipitate on
addition of excess ammonia solution. AgCl dissolves in
aqueous ammonia due to the formation of water woluble
One mole of the complex compound 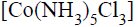 gives 3 moles of ions on dissolution in water. One mole of the same complex reacts with two moles of
gives 3 moles of ions on dissolution in water. One mole of the same complex reacts with two moles of 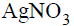 solution to yield two moles of AgCl(s).The structure of the complex is
solution to yield two moles of AgCl(s).The structure of the complex is
Which of the following compounds on heating at about
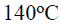 gives acetic acid ?
gives acetic acid ?
In which of the following reactions does
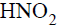 act as oxidizing agent ?
act as oxidizing agent ?
A compound of Xe and F is found to have 53.5% Xe. What is the oxidation number of Xe in this compound? (At. wt. of Xe = 131):-
Detailed Solution: Question 21
Detailed Solution: Question 22
Amongst the following, the total number of nucleophiles is -
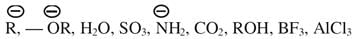
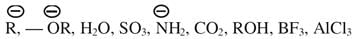
Detailed Solution: Question 23
How many of the following give positive isocyanide test
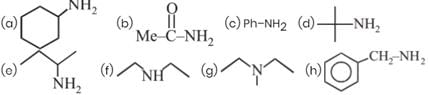

Detailed Solution: Question 25
1 videos|238 docs|217 tests |