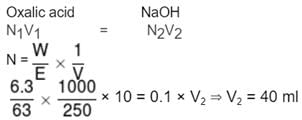JEE Main Chemistry Test- 2 Free Online Test 2026
MCQ Practice Test & Solutions: JEE Main Chemistry Test- 2 (25 Questions)
You can prepare effectively for JEE Mock Tests for JEE Main and Advanced 2026 with this dedicated MCQ Practice Test (available with solutions) on the important topic of "JEE Main Chemistry Test- 2". These 25 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 60 minutes
- - Number of Questions: 25
Sign up on EduRev for free to attempt this test and track your preparation progress.
Which of the following alkenes will react fastest with 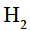 under catalytic hydrogenation conditions
under catalytic hydrogenation conditions
Detailed Solution: Question 1
The heat of hydrogenation of 1-hexene is 126 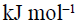 . When a second double bond is introduced in the molecule, the heat of hydrogenation of the resulting compound is 230
. When a second double bond is introduced in the molecule, the heat of hydrogenation of the resulting compound is 230 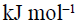 . The resulting compound (diene) is
. The resulting compound (diene) is
Detailed Solution: Question 2
The enthalpy of fusion of water is 1.435 kcal/mol. The molar entropy change for the melting of ice at 0°C is
Detailed Solution: Question 3
When 0.532 g of benzene (B.P. 80.°) is burnt in a constant volume system with an excess of oxygen, 22.3 KJ of heat is given out. ΔH for the combustion process is given by :
Detailed Solution: Question 4
The heat of formation of Al2O3 is 380 kcals/mole and that of Fe2O3 is 195 kcals/mole. The heat (in kcals/mole) of the thermite reaction is
Detailed Solution: Question 5
In the gaseous equilibrium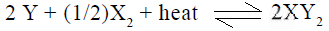 the formation of
the formation of 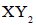 will be favored by
will be favored by
Detailed Solution: Question 6
The Kp for the decomposition of SO2Cl2 (if its degree of dissociation under one atomic pressure is 90%) is
Detailed Solution: Question 7
Detailed Solution: Question 8
If the maximum concentration of PbCl2 in water is 0.01M at 298K, its maximum concentration in 0.1M NaCl will be
Detailed Solution: Question 9
In a reversible chemical reaction at equilibrium, if the concentration of any one of the reactants is doubled, then the equilibrium constant will
Detailed Solution: Question 10
Ammonium hydrogen sulphide is contained in a closed vessel at 313 K when total pressure at equilibrium is found to be 0.8 atm. The value of Kp for the reaction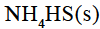

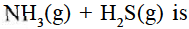
Detailed Solution: Question 11
Reactivity of hydrogen atoms attached to different carbon atoms in alkanes has the order
Detailed Solution: Question 12
Detailed Solution: Question 13
The major product obtained when isobutane is treated with chlorine in the presence of light is
Detailed Solution: Question 14
The compound contains atoms X,Y,Z. The oxidation number of X is 2, Y is 5 and Z is –2, a possible formula of the compound is
Detailed Solution: Question 15
Detailed Solution: Question 16
Enthalpy of neutralization of H3PO3 acid is –106.68 KJ/mole using NaOH. If enthalpy of neutralization of HCl by NaOH is –55.84 KJ/mole. Calculate ΔHionisation of H3PO3 into its ions. (in KJ)
Detailed Solution: Question 17
An aqueous solution of 6.3 g of oxalic acid dihydrate is made upto 250 mL. The volume of0.1 N NaOH required to completely neutralise 10 mL of this solution is :
Detailed Solution: Question 18
Detailed Solution: Question 19
100 mL of 0.6 M acetic acid is shaken with 2 g activated carbon. The final concentration of the solution after adsorption is 0.5 M. What is the amount of acetic acid adsorbed per gram of carbon.
Detailed Solution: Question 20
Which of the following is the most stable carbocation?
Detailed Solution: Question 21
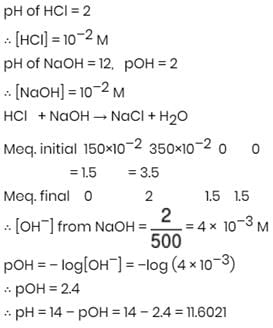
What will be the resultant pH when 150 mL of an aqueous solution of HCl (pH = 2.0) is mixed with 350 mL of an aqueous solution of NaOH (pH = 12.0)?
Detailed Solution: Question 22
Detailed Solution: Question 23
How many grams of NH4Cl should be dissolved per litre of solution to have a pH of 5.13 ? Kb for NH3 is 1.8 × 10–5.
Detailed Solution: Question 24
The oxidation state of chromium in potassium dichromate (K₂Cr₂O₇) is:
Detailed Solution: Question 25
360 docs|100 tests |