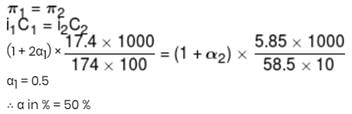JEE Main Chemistry Test- 4 Free Online Test 2026
MCQ Practice Test & Solutions: JEE Main Chemistry Test- 4 (25 Questions)
You can prepare effectively for JEE Mock Tests for JEE Main and Advanced 2026 with this dedicated MCQ Practice Test (available with solutions) on the important topic of "JEE Main Chemistry Test- 4". These 25 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 60 minutes
- - Number of Questions: 25
Sign up on EduRev for free to attempt this test and track your preparation progress.
An organic compound ‘X’ on treatment with acidified 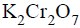 gives a compound ‘Y’ which reacts with
gives a compound ‘Y’ which reacts with 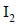 and sodium carbonate to form tri-iodomethane. The compound ‘X’ is
and sodium carbonate to form tri-iodomethane. The compound ‘X’ is
n-Propyl alcohol and iso-propyl alcohol can be chemically distinguished by which reagent
In the reaction of phenol with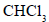 and aqueous NaOH at (343 K), the electrophile attacking the ring is
and aqueous NaOH at (343 K), the electrophile attacking the ring is
The compound which gives the most stable carbonium ion on dehydration is
The compound obtained by the reaction of ethene with diborane followed by hydrolysis with alkaline hydrogen peroxide is
When benzenediazonium chloride is treated with water, the compound formed is
What amount of bromine will be required to convert 2g of phenol into 2,4, 6-tribromophenol?
Detailed Solution: Question 13
The reaction of 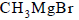 with acetone and hydrolysis of the resulting product gives
with acetone and hydrolysis of the resulting product gives
Which one of the following pairs will not form an ideal solution ?
Which of the following pairs of solutions is isotonic ?
At 298K, highest osmotic pressure is exhibited by a 0.1M solution of
The van’t Hoff factor for a 0.1 M 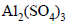 solution is 4.20. The degree of dissociation is
solution is 4.20. The degree of dissociation is
The molal boiling-point constant for water is 0.513 K/m When 0.1 mole of sugar is dissolved in 200.0 g of water, the solution boils under a pressure of 1.0 atm at
17.4 % (mass / volume) K2SO4 solution at 27°C is isotonic to 5.85% (mass / volume) NaCl solution at 27°C. If NaCl is 100% ionised, what is % ionisation of K2SO4 in aq. solution?
Detailed Solution: Question 21
Among the following ore how many example of carbonate ore:
lime stone, malachite, dolomite, Horn silver, Flourspar, Calamine, Cuprite.
lime stone, malachite, dolomite, Horn silver, Flourspar, Calamine, Cuprite.
Identify the element for given which have the most negative electron gain enthalpy. Write atomic number of that element as answer :-
P , S, Cl, F, Br
P , S, Cl, F, Br
Element 'X' has maximum electron affinity in periodic table then find the number of elements which have low first ionisation energy as compared to element 'X' ?
F, Na, S, Si, P, Ar
F, Na, S, Si, P, Ar
If number of low spin complexes are 'X' then find the value of 1/x.
[Co(NH3)6]+3, [CoF6]–3, [Fe(CN)6]–4, [Fe(H2O)6]+2, [Cr(gly)3]0, [CoBr2Cl2]–2
[Co(NH3)6]+3, [CoF6]–3, [Fe(CN)6]–4, [Fe(H2O)6]+2, [Cr(gly)3]0, [CoBr2Cl2]–2
360 docs|100 tests |