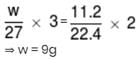JEE Main & Advanced Chemistry Test- 7 Free Online Test 2026
MCQ Practice Test & Solutions: JEE Main Chemistry Test- 7 (25 Questions)
You can prepare effectively for JEE JEE Main & Advanced Mock Test Series 2026 with this dedicated MCQ Practice Test (available with solutions) on the important topic of "JEE Main Chemistry Test- 7". These 25 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 60 minutes
- - Number of Questions: 25
Sign up on EduRev for free to attempt this test and track your preparation progress.
To which class of compounds do enzymes usually belong ?
Detailed Solution: Question 1
XeF4 is prepared by heating Xe and F2 at 400oC in the ratio of
Glyptal, a highly cross-linked rigid thermosetting resin,is prepared by heating
The polymer obtained by condensation polymerization is
Which of the following polymer type is not classified on the basis of its application and properties?
Detailed Solution: Question 18
The colour of the precipitate formed when a reducing sugar is heated with Fehling’s solution is
On passing electric current through moten AlCl3, 11.2 litre of Cl2 is liberated at N.T.P. at anode. The quantity of aluminium deposited at cathode in g will be :- (At. wt. of Al = 27)
Detailed Solution: Question 21
How many of the following are condensation copolymers ?
Nylon-6, nylon-66, Dacron, glyptal, buna-S, ABS, neoprene, PHBV, perlon-U.
Nylon-6, nylon-66, Dacron, glyptal, buna-S, ABS, neoprene, PHBV, perlon-U.
Detailed Solution: Question 22
A 250.0 ml sample of a 0.20 M Cr3+ is electrolyzed with a current of 96.5 A , so as to deposit Cr. If the remaining [Cr3+] is 0.1M, the time taken (in sec.) in the process.
Detailed Solution: Question 23
How many statements are correct in the following -
(a) Reduction potential of Pb2+ is more than Zn2+
(b) Mercury cell is an example of primary cell
(c) Hydrogenation of ethene (C2H4) is a first order reaction
(d) order of wavelength γ-rays < X-ray < IR
(e) Molal elevation constant (Kb) of ethanol is more than carbon tetrachloride (CCl4)
(f) Sols of starch, gum, gelatin are postively charged.
(g) In coagulation of positive sol, the flocculating power is in order
: [Fe(CN)6]–4 > PO4–3 > SO4–2 > Cl–
(h) In Rhombohedral unit cell : a = b = c
(a) Reduction potential of Pb2+ is more than Zn2+
(b) Mercury cell is an example of primary cell
(c) Hydrogenation of ethene (C2H4) is a first order reaction
(d) order of wavelength γ-rays < X-ray < IR
(e) Molal elevation constant (Kb) of ethanol is more than carbon tetrachloride (CCl4)
(f) Sols of starch, gum, gelatin are postively charged.
(g) In coagulation of positive sol, the flocculating power is in order
: [Fe(CN)6]–4 > PO4–3 > SO4–2 > Cl–
(h) In Rhombohedral unit cell : a = b = c
Find out the ratio of incorrect and correct statements about H2O2 among the following?
1. In the pure state H2O2 is almost colorless (very pale blue)
2. Hydrogen peroxide has non planar structure in both gas phase & solid phase
3. 2-ethylanthraquinol react with water to give H2O2
4. H2O2 is used in pollution control
5. Dihedral angle of H2O2 is larger in gas phase compared to that in solid phase
1. In the pure state H2O2 is almost colorless (very pale blue)
2. Hydrogen peroxide has non planar structure in both gas phase & solid phase
3. 2-ethylanthraquinol react with water to give H2O2
4. H2O2 is used in pollution control
5. Dihedral angle of H2O2 is larger in gas phase compared to that in solid phase
Detailed Solution: Question 25
1 videos|238 docs|217 tests |