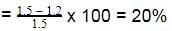Thermochemistry - Free MCQ Practice Test with solutions, NEET
MCQ Practice Test & Solutions: Test: Thermochemistry (30 Questions)
You can prepare effectively for NEET Topic-wise MCQ Tests for NEET with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Thermochemistry". These 30 questions have been designed by the experts with the latest curriculum of NEET 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 45 minutes
- - Number of Questions: 30
Sign up on EduRev for free to attempt this test and track your preparation progress.
Detailed Solution: Question 1
Detailed Solution: Question 2
How much heat will be required at constant pressure to form 1.28 kg of CaC2 from CaO(s) & C(s) ?
Given :
ΔfH°(CaO, s) = -152 kcal/mol
ΔfH°(CaC2, s) = -14 kcal/mol
ΔfH°(CO, g) = -26 kcal/mol
Detailed Solution: Question 3
50.0 mL of 0.10 M HCl is mixed with 50.0 mL of 0.10 M NaOH. The solution temperature rises by 3.0°C Calculate the enthalpy of neutralization per mole of HCl. [take proper assumptions]
Detailed Solution: Question 4
The enthalpy of neutralisation of a weak acid in 1 M solution with a strong base is -56.1 kJ mol-1. If the enthalpy of ionization of the acid is 1.5 kJ mol-1 and enthalpy of neutralization of the strong acid with a strong base is -57.3 kJ equiv-1, what is % ionization of the weak acid in molar solution (assume the acid to be monobasic) ?
Detailed Solution: Question 5
For the allotropic change represented by the equation C (graphite) → C (diamond), ΔH = 1.9 kJ. If 6 g of diamond and 6 g of graphite are separately burnt to yield CO2, the heat liberated in first case is
Detailed Solution: Question 6
If x1, x2 and x3 are enthalpies of H - H, O = O and O - H bonds respectively, and x4 is the enthalpy of vaporisation of water, estimate the standard enthalpy of combustion of hydrogen
Detailed Solution: Question 7
NH3(g) + 3Cl2(g) 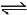 NCl3(g) + 3HCl(g) ; -ΔH1
NCl3(g) + 3HCl(g) ; -ΔH1
N2(g) + 3H2(g) 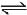 2NH3(g) ; ΔH2
2NH3(g) ; ΔH2
H2(g) + Cl2(g) 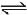 2HCl(g) ; ΔH3
2HCl(g) ; ΔH3
The heat of formation of NCl3 (g) in the terms of ΔH1, ΔH2 and ΔH3 is
Detailed Solution: Question 8
The enthalpy of neutralisation of HCl and NaOH is -57 kJ mol-1. The heat evolved at constant pressure (in kJ) when 0.5 mole of H2SO4 react with 0.75 mole of NaOH is equal to
Detailed Solution: Question 9
Reaction involving gold have been of particular interest to a chemist. Consider the following reactions.
Au(OH)3 + 4 HCl → HAuCl4 + 3H2O, ΔH = -28 kcal
Au(OH)3 + 4 HBr → HAuBr4 + 3 H2O, ΔH = -36.8 kcal
In an experiment there was an absorption of 0.44 kcal when one mole of HAuBr4 was mixed with 4 moles of HCl. What is the percentage conversion of HAuBr4 into HAuCl4 ?
Detailed Solution: Question 10
(i) Cis - 2 - butene → trans - 2 - butene, ΔH1
(ii) Cis - 2 - butene → 1 - butene, ΔH2
(iii) Trans - 2 - butene is more stable than cis - 2 - butene.
(iv) Enthalpy of combustion of 1 - butene, ΔH = -649.8 kcal/mol
(v) 9ΔH1 + 5 ΔH2 = 0
(vi) Enthalpy of combustion of trans 2 - butene, ΔH = -647.0 kcal/mol
Q. The value of ΔH1 & ΔH2 in Kcal/mole are
Detailed Solution: Question 11
The reaction CH4(g) + Cl2(g) → CH3Cl(g) + HCl(g) has ΔH = -25 kCal.
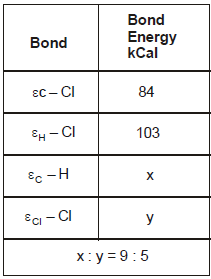
From the given data, what is the bond energy of Cl - Cl bond
Detailed Solution: Question 12
From the following data at 25°C
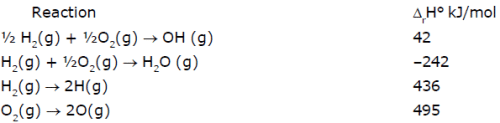
Which of the following statement(s) is/are correct :
Statement(a) : ΔrH° for the reaction H2O(g) → 2H(g) + O(g) is 925 kJ/mol
Statement(b) : ΔrH° for the reaction OH(g) → H(g) + O(g) is 502 kJ/mol
Statement(c) : Enthalpy of formation of H(g) is-–218 kJ/mol
Statement(d) : Enthalpy of formation of OH(g) is 42 kJ/mol
Statement(a) : ΔrH° for the reaction H2O(g) → 2H(g) + O(g) is 925 kJ/mol
Statement(b) : ΔrH° for the reaction OH(g) → H(g) + O(g) is 502 kJ/mol
Statement(c) : Enthalpy of formation of H(g) is-–218 kJ/mol
Statement(d) : Enthalpy of formation of OH(g) is 42 kJ/mol
Detailed Solution: Question 13
The standard molar enthalpies for formation of cyclohexane (l) & benzene (l) at 25°C are -156 & + 49 kJ/mol respectively. The standard enthalpy of hydrogenation of cyclohexane (l) at 25° is -119 kJ mol-1. Use these data to estimate the magnitude of the resonance energy of benzene.
For hypothetical reaction -
A(g) + B (g) → C (g) + D (g)
Which of the following statements is correct -
ΔH for CaCO3(s) → CaO(s) + CO2(g) is 176 kJ mol-1 at 1240 K. The ΔU for the change is equal to :
Detailed Solution: Question 16
Detailed Solution: Question 17
When enthalpy of reactants is higher than product then reaction will be
Detailed Solution: Question 18
In the combustion of 4g. of CH4, 2.5 K cal of heat is liberated. The heat of combustion of CH4 is -
Detailed Solution: Question 19
Ammonium nitrate can decompose with explosion by the following reaction.
NH4NO3 (s) → N2O (g) + 2H2O ;
ΔH = -37.0 KJ/mol
Calculate the heat produced when 2.50g of NH4NO3 decomposes -
Detailed Solution: Question 20
From the following data, the heat of formation of Ca(OH)2(s) at 18°C is ………..kcal:
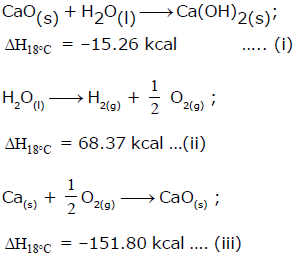
Detailed Solution: Question 21
Detailed Solution: Question 23
Detailed Solution: Question 24
HA + OH- → H2O + A- + q1 kJ
H+ + OH- → H2O + q2 kJ
The enthalpy of dissociation of HA is
Detailed Solution: Question 25
The value of ΔHsol. of BaCl2(s) and BaCl2. 2H2O (s) are – a kJ and b kJ respectively. The value of ΔH Hydration of BaCl2 (s) is-
Detailed Solution: Question 26
A solution of 500 ml of 0.2 M KOH and 500 ml of 0.2 M HCl is mixed and stirred; the rise in temperature is T1. The experiment is repeated using 250 ml of each solution, the temperature raised is T2. Which of the following is true -
Detailed Solution: Question 27
The net heat change in a chemical reaction is same whether it is brought about in two or more different ways in one or several steps. It is known as -
Detailed Solution: Question 28
According to Hess's Law the thermal effect of a reaction depends on -
Detailed Solution: Question 29
How many kcal of heat is evolved by the complete neutralisation of one mole sulphuric acid with NaOH -
Detailed Solution: Question 30
6 docs|803 tests |