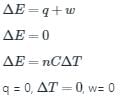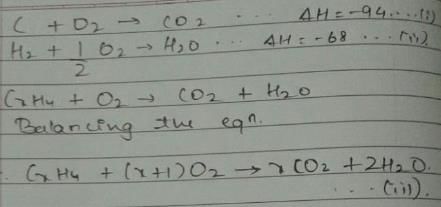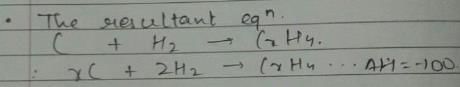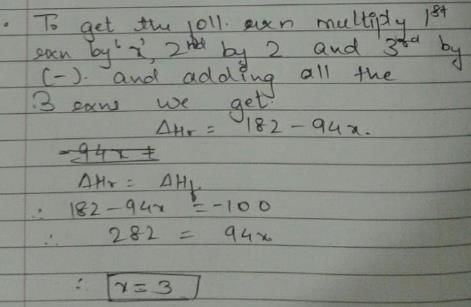Hess's Law of Constant Heat Summation - Free MCQ Practice Test with solutions,
MCQ Practice Test & Solutions: Test: Hess's Law of Constant Heat Summation (15 Questions)
You can prepare effectively for NEET Topic-wise MCQ Tests for NEET with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Hess's Law of Constant Heat Summation". These 15 questions have been designed by the experts with the latest curriculum of NEET 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 15 minutes
- - Number of Questions: 15
Sign up on EduRev for free to attempt this test and track your preparation progress.
Direction (Q. Nos. 1-7) This section contains 7 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONLY ONE option is correct.
Given,
The enthalpies of elements in their standard states are taken as zero. The enthalpy of formation of a compound
Detailed Solution: Question 1
Detailed Solution: Question 2
Diborane is a potential rocket fuel which undergoes combustion according to the reaction,
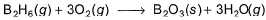
From the following data, enthalpy change for the combustion of diborane is
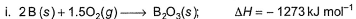
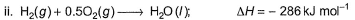

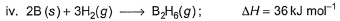
Detailed Solution: Question 3
Following diagram represents Born-Haber cycle to determine lattice energy of NaCI(s). It is based on Hess’s law of constant heat summation. ΔlatticeH° of
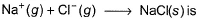
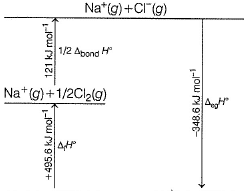
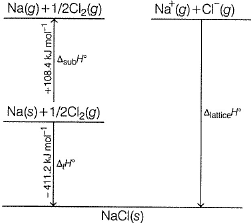
Detailed Solution: Question 4
Detailed Solution: Question 5
Given, HCI (g) → H (g) + Cl (g) at 298 K (say temperature T K),

For
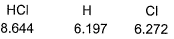
Q. Thus, for the given reaction  (at 0 K) is
(at 0 K) is
Detailed Solution: Question 6
The measured enthalpy change for burning of ketene (g) (CH2CO) is - 981.1 kJ mo-1 at 298 K ,CH2CO (g)+ 2O2(g) → 2CO2 (g) + H2O (g) and that of CH4 (g) is - 802.3 kJ mol-1 at 298 K

Thus, enthalpy change at 298 K for the following thermochemical reaction is
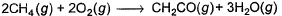
Detailed Solution: Question 7
The condition for free expansion of an ideal gas under adiabatic condition is
Detailed Solution: Question 8
Direction (Q. Nos. 9 and 10) This section contains a paragraph, wach describing theory, experiments, data etc. three Questions related to paragraph have been given.Each question have only one correct answer among the four given ptions (a),(b),(c),(d).
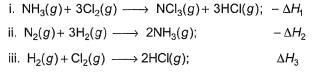
Consider the following thermochemical equations
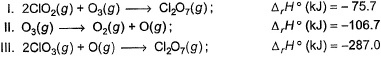
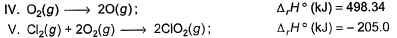
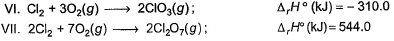
Q. ΔrH° of the following thermochemical reaction is
CIO2 (g) + O (g) → CIO3 (g)
Detailed Solution: Question 9
Consider the following thermochemical equations
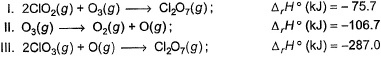
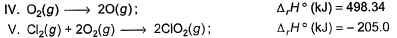
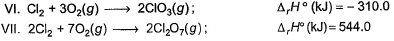
Q. ΔrH° of the following thermochemical reaction is
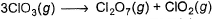
Direction (Q. Nos. 11) Choice the correct combination of elements and column I and coloumn II are given as option (a), (b), (c) and (d), out of which ONE option is correct.
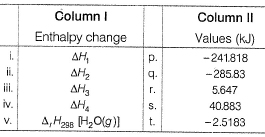
Q. For liquid water at
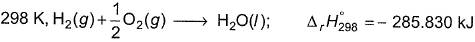

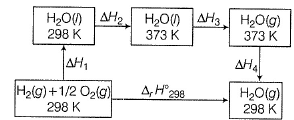
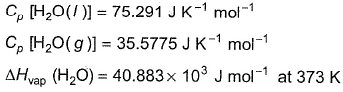
Compare the parameters ΔH1, ΔH2 ... in Column I with the corresponding values in kJ in Column II.

Detailed Solution: Question 11
Direction (Q. Nos. 12-15) This section contains 4 questions. when worked out will result in an integer from 0 to 9 (both inclusive).
Q. Based on (BE) values, ΔrH° of the following reaction 298 K is
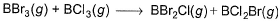
Detailed Solution: Question 12
Heat of combustion of CH2CO(g) is - 981.1 kJ mol-1 and that of CH4 (g) is - 802.3 kJat 298 K. If 1247.0 kJ of heat is released in the following change
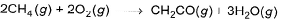
Q. How many moles of CH4(g)are used?
Detailed Solution: Question 13
Consider the following reactions
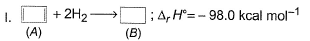
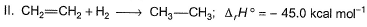
Q. What is the resonance energy (in kcal) of A?
Detailed Solution: Question 14
Given, enthalpy of combustion of carbon (s) = -94 kcal mol-1
H2(g) = - 68 kcal mol-1 and CXH4 = - 318 kcal mol-1 and ΔfH° (CxH4) = - 100 kcal mol-1
Q. What is the value of x?
Detailed Solution: Question 15
6 docs|803 tests |