Thermodynamics - Free Assertion & Reason Questions with Solutions NEET
MCQ Practice Test & Solutions: Assertion & Reason Test: Thermodynamics (8 Questions)
You can prepare effectively for NEET NCERT Based Tests for NEET with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Assertion & Reason Test: Thermodynamics". These 8 questions have been designed by the experts with the latest curriculum of NEET 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 16 minutes
- - Number of Questions: 8
Sign up on EduRev for free to attempt this test and track your preparation progress.
In the following questions, a statement of Assertion (A) followed by a statement of Reason (R) is given. Choose the correct option out of the choices given below each question.
Assertion (A): Combustion of all organic compounds is an exothermic reaction.
Reason (R) : The enthalpies of all elements in their standard state are zero.
Detailed Solution: Question 1
In the following questions, a statement of Assertion (A) followed by a statement of Reason (R) is given. Choose the correct option out of the choices given below each question.
Assertion (A) : Spontaneous process is an irreversible process and may be reversed by some external agency.
Reason (R) : Decrease in enthalpy is a contributory factor for spontaneity.
Detailed Solution: Question 2
In the following questions, a statement of Assertion (A) followed by a statement of Reason (R) is given. Choose the correct option out of the choices given below each question.
Assertion (A) : A liquid crystallises into a solid and is accompanied by decrease in entropy.
Reason (R) : In crystals, molecules organise in an ordered manner.
Detailed Solution: Question 3
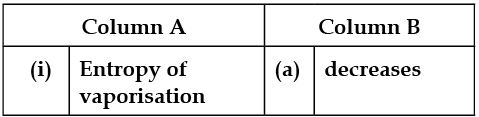
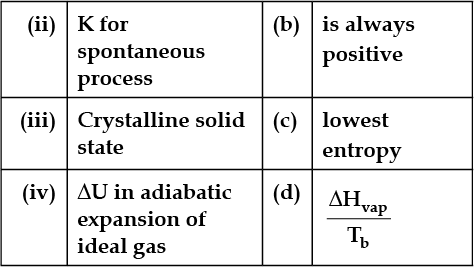
In the following questions, more than one correlation is possible between options of both columns.
Match the following:


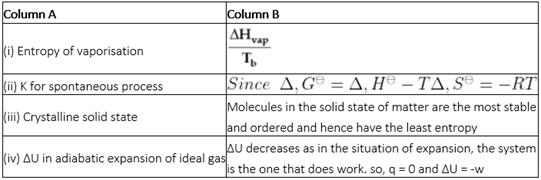
Detailed Solution: Question 4
Match the following processes with entropy change:


Detailed Solution: Question 5
Match the following parameters with description for spontaneity :

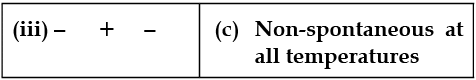

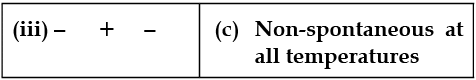
Detailed Solution: Question 6
Detailed Solution: Question 7
Assertion: The second law of thermodynamics states that the entropy of a closed or isolated system always increases. This means that all available energy is used up and there is no more potential for further useful work.
Reason: The system becomes disordered and also degraded.
Detailed Solution: Question 8