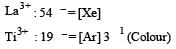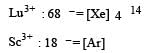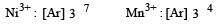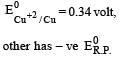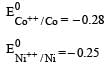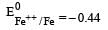31 Year NEET Previous Questions: The d & f-Block Elements - 1 - Free MCQ
MCQ Practice Test & Solutions: 31 Year NEET Previous Year Questions: The d & f-Block Elements - 1 (35 Questions)
You can prepare effectively for NEET Chemistry Class 12 with this dedicated MCQ Practice Test (available with solutions) on the important topic of "31 Year NEET Previous Year Questions: The d & f-Block Elements - 1". These 35 questions have been designed by the experts with the latest curriculum of NEET 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 70 minutes
- - Number of Questions: 35
Sign up on EduRev for free to attempt this test and track your preparation progress.
Which of the following statement is not correct? [2001]
Detailed Solution: Question 1
General electronic configuration of lanthanides is[2002]
Detailed Solution: Question 2
Which of the following shows maximum number of oxidation states? [2002]
Detailed Solution: Question 3
The basic character of the transition metal monoxides follows the order [2003] (Atomic Nos.,Ti = 22, V = 23, Cr = 24, Fe = 26)
Detailed Solution: Question 4
Which one of the following characteristics of the transition metals is associated with their catalytic activity? [2003]
Detailed Solution: Question 5
The correct order of ionic radii of Y3+, La3+, Eu3+ and Lu3+ is [2003]
Detailed Solution: Question 6
Among the following series of transition metal ions, the one where all metal ions have 3d2 electronic configuration is(At. nos. Ti = 22; V = 23; Cr = 24; Mn = 25) [2004]
Detailed Solution: Question 7
Lanthanoids are [2004]
Detailed Solution: Question 8
The aqueous solution containing which one of the following ions will be colourless? (Atomic number: Sc = 21, Fe = 26, Ti = 22, Mn = 25)[2005]
Detailed Solution: Question 9
The main reason for larger number of oxidation states exhibited by the actinoids than the corresponding lanthanoids, is [2005, 2006]
Detailed Solution: Question 10
Four successive members of the first row transition elements are listed below with their atomic numbers. Which one of them is expected to have the highest third ionization enthalpy? [2 00 5]
Detailed Solution: Question 11
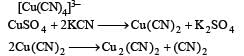
Copper sulphate dissolves in excess of KCN to give[2006]
Detailed Solution: Question 12
In which of the following pairs are both the ions coloured in aqueous solutions ? [2006] (At. no. : Sc = 21, Ti = 22, Ni = 28, Cu = 29, Co = 27)
Detailed Solution: Question 13
Which of the following oxidation states are the most characteristic for lead and tin respectively?
Detailed Solution: Question 14
Which one of the following ions is the most stable in aqueous solution? [2007] (At.No. Ti = 22, V = 23, Cr = 24, Mn = 25)
Detailed Solution: Question 15
Identify the incorrect statement among the following: [2007]
Detailed Solution: Question 16
The correctorder of decreasing second ionisation enthalpy of Ti (22), V(23), Cr(24) and Mn (25) is : [2008]
Detailed Solution: Question 17
Which one of the elements with the following outer orbital configurations may exhibit the largest number of oxidation states? [2009]
Detailed Solution: Question 18
Qut of TiF62–, CoF63–, Cu2Cl2 and NiCl24– (Z of Ti = 22, Co = 27, Cu = 29, Ni = 28), the colourless species are: [2009]
Detailed Solution: Question 19
Which of the following ions will exhibit colour in aqueous solutions? [2010]
Detailed Solution: Question 20
Which one of the following ions has electronic configuration [Ar] 3d6 ? [2010]
(At. Nos. Mn = 25, Fe = 26, Co = 27, Ni = 28)
Detailed Solution: Question 21
Which of the following pairs has the same size?
Detailed Solution: Question 22
Which of the following oxidation states is the most common among the lanthanoids? [2010]
Detailed Solution: Question 23
For the four successive transition elements (Cr, Mn, Fe and Co), the stability of +2 oxidation state will be there in which of the following order?
Detailed Solution: Question 24
Acidified K2Cr2O7 solution turns green when Na2SO3 is added to it. This is due to the formation of : [2011]
Detailed Solution: Question 25
Which of the statements is not true? [2012]
Detailed Solution: Question 26
Which one of the following does not correctly represent the correct order of the property indicated against it?
1. Ti < V < Cr < Mn : increasing number of oxidation states
2. Ti3+ < V3+ < Cr3+ < Mn3+ : increasing magnetic moment
3. Ti < V < Cr < Mn : increasing melting points
4. Ti < V < Mn < Cr : increasing 2nd ionization enthalpy
Detailed Solution: Question 27
Four successive members of the first series of the transition metals are listed below. For which one of them the standard potential (E0M2+ /M) value has a positive sign? [2012 M]
Detailed Solution: Question 28
The catalytic activity of transition metals and their compounds is ascribed mainly to :[2012 M]
Detailed Solution: Question 29
Which of the following exhibit only + 3 oxidation state ? [2012 M]
Detailed Solution: Question 30
54 videos|290 docs|74 tests |



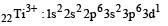
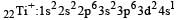
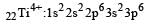
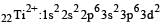
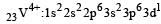 Thus option (b) is discarded
Thus option (b) is discarded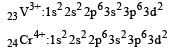
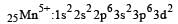
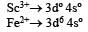

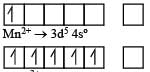
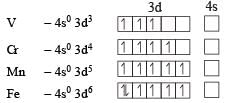

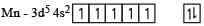
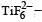 Ti is in + 4 O.S. ; 3d0 = colourless
Ti is in + 4 O.S. ; 3d0 = colourless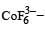 Co is in + 3 O.S ; 3d5 = coloured
Co is in + 3 O.S ; 3d5 = coloured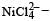 Ni is in + 2 O.S ; 3d8 – coloured
Ni is in + 2 O.S ; 3d8 – coloured