Chemistry - (CY) 2018 GATE Paper (Practice Test) - GATE MCQ
30 Questions MCQ Test - Chemistry - (CY) 2018 GATE Paper (Practice Test)
“When she fell down the _______, she received many _______ but little help.”
The words that best fill the blanks in the above sentence are
“In spite of being warned repeatedly, he failed to correct his _________ behaviour.”
The word that best fills the blank in the above sentence is
| 1 Crore+ students have signed up on EduRev. Have you? Download the App |
For 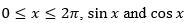 are both decreasing functions in the interval ________.
are both decreasing functions in the interval ________.
The area of an equilateral triangle is √3. What is the perimeter of the triangle?
Arrange the following three-dimensional objects in the descending order of their volumes:
(i) A cuboid with dimensions 10 cm, 8 cm and 6 cm
(ii) A cube of side 8 cm
(iii) A cylinder with base radius 7 cm and height 7 cm
(iv) A sphere of radius 7 cm
An automobile travels from city A to city B and returns to city A by the same route. The speed of the vehicle during the onward and return journeys were constant at 60 km/h and 90 km/h, respectively. What is the average speed in km/h for the entire journey?
A set of 4 parallel lines intersect with another set of 5 parallel lines. How many parallelograms are formed?
To pass a test, a candidate needs to answer at least 2 out of 3 questions correctly. A total of 6,30,000 candidates appeared for the test. Question A was correctly answered by 3,30,000 candidates. Question B was answered correctly by 2,50,000 candidates. Question C was answered correctly by 2,60,000 candidates. Both questions A and B were answered correctly by 1,00,000 candidates. Both questions B and C were answered correctly by 90,000 candidates. Both questions A and C were answered correctly by 80,000 candidates. If the number of students answering all questions correctly is the same as the number answering none, how many candidates failed to clear the test?
If �� 2 + �� − 1 = 0 what is the value of
In a detailed study of annual crow births in India, it was found that there was relatively no growth during the period 2002 to 2004 and a sudden spike from 2004 to 2005. In another unrelated study, it was found that the revenue from cracker sales in India which remained fairly flat from 2002 to 2004, saw a sudden spike in 2005 before declining again in 2006. The solid line in the graph below refers to annual sale of crackers and the dashed line refers to the annual crow births in India. Choose the most appropriate inference from the above data.
The major product formed in the following reaction is
The major product formed in the following reaction is
The major product of the following intramolecular cycloaddition reaction is
The major product of the following reaction is
The major product formed in the following reaction sequence is
The major product formed in the following reaction sequence is
The spherical harmonic function, with appropriate values of l and m, is an eigenfunction of
operator. The corresponding eigenvalue is
Consider the operators, are the position and linear momentum operators, respectively. The commutator,
is equal to
The temperature derivative of electrochemical cell potential E at constant pressure,is given by
For an ionic micelle-forming surfactant near its critical micelle concentration (CMC), the dependence of molar conductivity and surface tension on surfactant concentration is best represented by
According to Eyring transition state theory for a bimolecular reaction, the activated complex has
Based on Wade’s rule, the structure-type of [B5H8]– is
The coordination geometries around the copper ion of plastocyanin (a blue-copper protein) in oxidized and reduced form, respectively, are
The water exchange rates for the complex ions follow the order
The lowest energy d → d transition of the complexes follow the order
The symmetry label of valence p orbitals of a metal ion in an octahedral ligand field is
The bond angle (Ti-C-C) in the crystal structure ofis severely distorted due to
The molar heat capacity of a substance is represented in the temperature range 298 K to 400 K by the empirical relation where b is a constant. The molar enthalpy change when the substance is heated from 300 K to 350 K is 2 kJ mol-1. The value of b is___________ J K-2 mol-1. (Up to two decimal places)
For low partial pressure of ozone (O3), the adsorption of ozone on graphite surface is fully dissociative in nature, and follows Langmuir isotherm. Under these conditions, if the dependence of the surface coverage of graphite (θ) on partial pressure of ozone ( PO3 ) is given by the value of x is___________. (Up to two decimal places)
For the radioactive isotope 131I, the time required for 50% disintegration is 8 days. The time required for the 99.9% disintegration of 5.5 g of 131I is ________ days. (Up to one decimal place)

















