Heterocyclic Chemistry & Natural Process - GATE Chemistry MCQ
20 Questions MCQ Test GATE Chemistry Mock Test Series - Heterocyclic Chemistry & Natural Process
Which of the following test is used for the detection of aldoses?
| 1 Crore+ students have signed up on EduRev. Have you? Download the App |
The enzyme which cuts from the N-terminal side of the amino acids having Leu, Asp and Glu amino acids is/are
The protecting group of amines which is deprotected by base is
The number of π-electrons which are in delocalization in pyridine ring is/are____
Among the following sugars which gives both Bromine water and Tollen’s Test positive is/are
D-glucose, D-Fructose, D-mannose, D-iodose
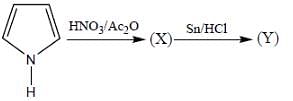
The most reactive position for the nitration in the following compounds is
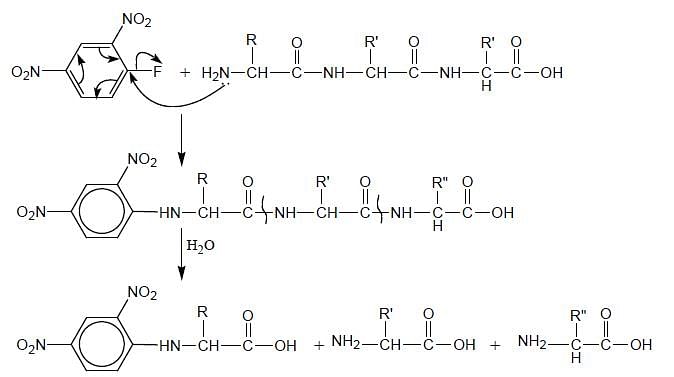
If a tripeptide is treated with Sangers reagent followed by hydrolysis. How many amide bonds will be hydrolysed in a peptide chain?
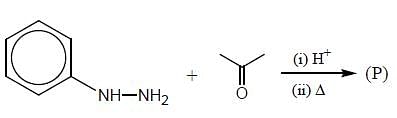
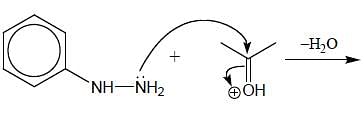
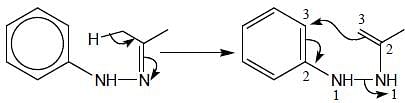
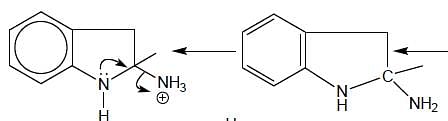
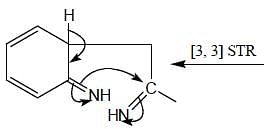
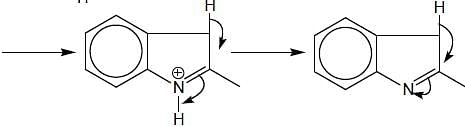
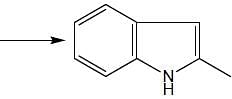
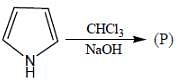
In the following reaction, how many membered ring will be formed as major product
Among the following amino acids, how many amino acids having a sulfur groups
(I) Cysteine (II) Methionine (III) Arginine (IV) Valine (V) Lyscine (VI) Cystine
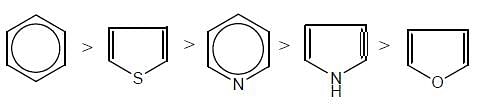
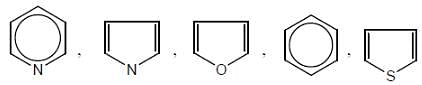
Give the aromaticity order in the following compounds
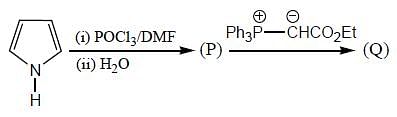
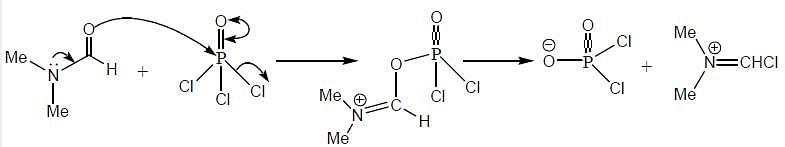
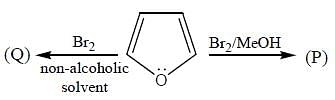
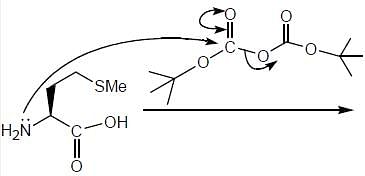
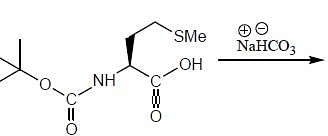
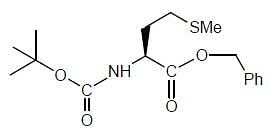
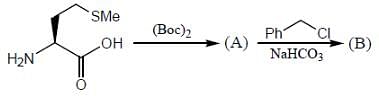
The products (A) and (B) formed in the following reaction sequence are
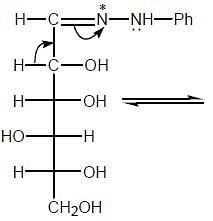
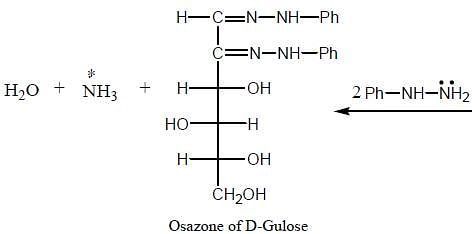
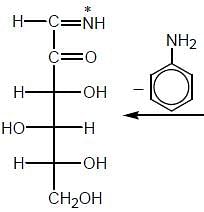
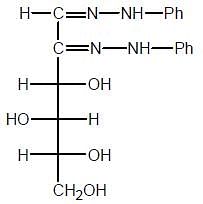
The osazone (A) could be obtained from :
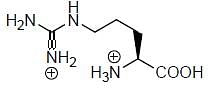
Among the following amino acids, how many is/are basic
Val, Arg, His, Cys, Leu, Lys, Met
Calculate the isoelectronic point value of the salt of Argnine, having pKa values; 10.9, 8.5, 2.2, respectively.
|
18 docs|37 tests
|
|
18 docs|37 tests
|


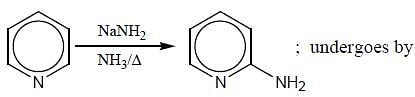
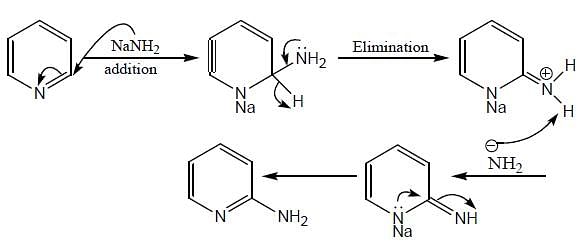
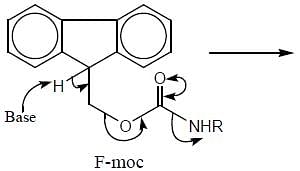
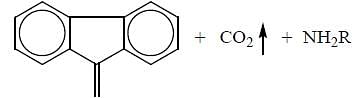
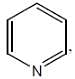
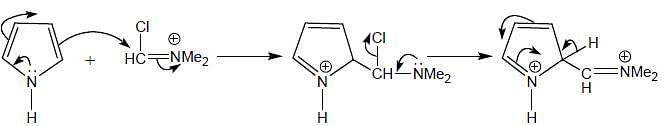
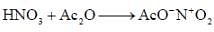 (mild nitrating agent) electrophilic addition on pyrrol takes place at second position then third
(mild nitrating agent) electrophilic addition on pyrrol takes place at second position then third 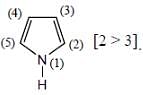 And Sn/HCI converts NO2 to NH2 group.
And Sn/HCI converts NO2 to NH2 group.