Olympiad Test: Changes Around Us - Class 6 MCQ
20 Questions MCQ Test - Olympiad Test: Changes Around Us
Raveena covered a beaker of boiled water with a metal tray as shown in the diagram.
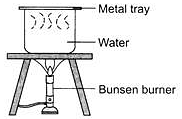
The beaker and metal tray were then removed from the tripod stand and left on the table. After two minutes, which of the following can be observed?

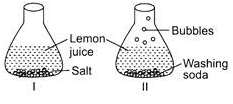
Sudha took lemon juice in two flasks. She put salt in flask I and washing soda in flask II. She observed lots of bubbles in flask II but nothing in flask I. What could be the reason for this?

| 1 Crore+ students have signed up on EduRev. Have you? Download the App |
Study the given Venn diagram carefully.
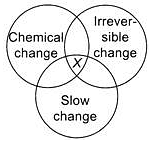
The centre point X represents

A metallic piece (X) is dropped into copper sulphate solution. After some time, the blue colour of the solution changes to green. The change which has occurred is

Read the given passage and fill in the blanks by choosing an appropriate option. Iron nails when new are grey and bright. When exposed to (i), they get covered with a (ii) coating. This process is called (iii) and it is a (iv) change.
Few changes are given below. Mark the correct option from the codes given below.
(i) Milk is set to form curd.
(ii) Batter is steamed to form idlis.
(iii) Water is boiled to form steam.
(iv) Salt is dissolved in water to form solution.
Nitu mixed some iron filings with sulphur powder in a China dish. She heated the contents of China dish. What did she observe during the experiment?
What is common among the following phenomena?
1. Blinking of traffic lights.
2. Rotation of blades of a fan.
3. Swinging of pendulum of a clock.
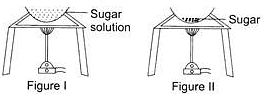
Observe the two figures given below. In figure I, a solution of sugar in water is heated and in figure II, only sugar is being heated. What is the correct sequence of changes taking place?
Rohan tore a sheet of paper into pieces and then burnt them. Identify the irreversible change taking place in the process.
Study the flowchart carefully.
(i) Melting of ice (i) Cutting of paper
(ii) Blowing of balloon (ii) Flowering of bud
Which of the following is correct?
A potter shapes pots out of clay (Process 1). He then bakes the pots in an oven (Process 2). Which of the following statements is correct?
Match the column I with column II and choose the correct option from the codes given below:
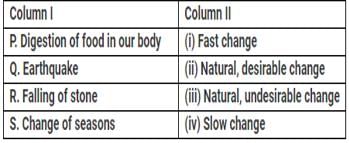
Consider the following phenomena:
(i) Forest fire
(ii) Growth of plants
(iii) Low and high tides
(iv) Occurrence of a rainbow
(v) Earthquakes
(vi) Opening of a morning glory flower.
Which of the above phenomena can be classified as periodic changes?
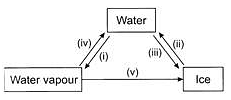
Observe the given figure carefully and choose the correct statement,
Swati classified a few changes occurring around us as shown in the table.
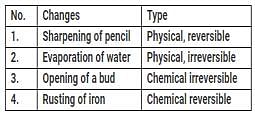
Which of the changes is/are classified correctly?
Read the given statements and mark the correct option.
Statement 1: While fixing a metal rim on a wooden wheel of a cart, the size of the metal rim is kept slightly smaller than that of the wooden wheel.
Statement 2: Metals expand on heating and contract on cooling.
Study the given Venn diagram.
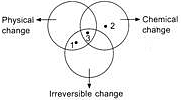
Identify points 1, 2 and 3.
Observe the given processes 1 and 2 carefully.
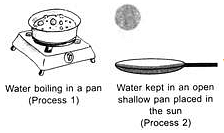
Which of the following statements are correct?
I. Bubbles are formed in process 1 while no bubble formation takes place in process 2.
II. Process 1 is slow while process 2 is fast.
Ill. Process 1 occurs at 100oC while process 2 can occur at any temperature.

















