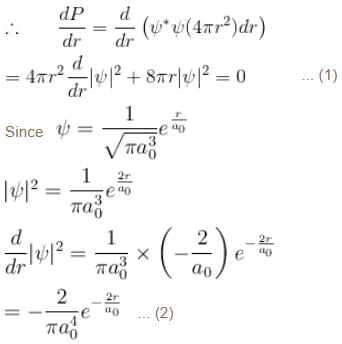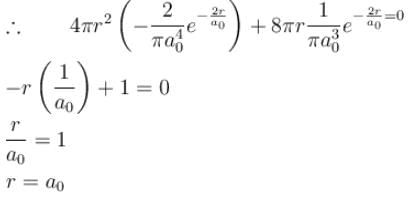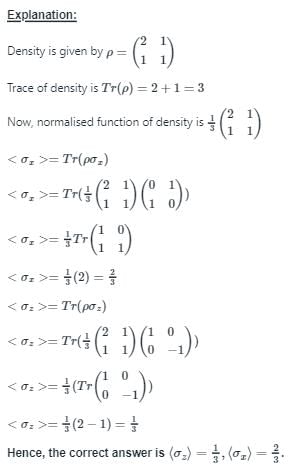Postulates Of Quantum Mechanics NAT - Free MCQ Test with solutions
MCQ Practice Test & Solutions: Postulates Of Quantum Mechanics NAT (10 Questions)
You can prepare effectively for Physics Topic wise Tests for IIT JAM Physics with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Postulates Of Quantum Mechanics NAT". These 10 questions have been designed by the experts with the latest curriculum of Physics 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 45 minutes
- - Number of Questions: 10
Sign up on EduRev for free to attempt this test and track your preparation progress.
The wave function 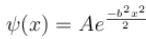 where A and B are real constant, is a normalised eigenfunction of the Schrödinger equation for a particle of mass M and energy E in a one dimensional potential V(x) such that V(x) = 0 at x = 0. Find the value for V in units of
where A and B are real constant, is a normalised eigenfunction of the Schrödinger equation for a particle of mass M and energy E in a one dimensional potential V(x) such that V(x) = 0 at x = 0. Find the value for V in units of 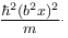
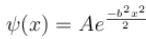 where A and B are real constant, is a normalised eigenfunction of the Schrödinger equation for a particle of mass M and energy E in a one dimensional potential V(x) such that V(x) = 0 at x = 0. Find the value for V in units of
where A and B are real constant, is a normalised eigenfunction of the Schrödinger equation for a particle of mass M and energy E in a one dimensional potential V(x) such that V(x) = 0 at x = 0. Find the value for V in units of 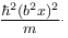
Detailed Solution: Question 1
In the spectrum of hydrogen, what is the ratio of the longest wavelength in the Lymann series to the longest is Balmer series ?
Detailed Solution: Question 2
In a photoelectric effect set up two metals with work function φ1 and φ2 are used. For the same value of incident frequency, the stopping potential for metal 1 is twice that for metal 2. If the incident energy is 5eV and the φ1 = 1.5eV. Find φ2. (in eV)
Detailed Solution: Question 3
A particle is in an infinite square well potential with walls at x = 0 and x = L. If the particle is in the state 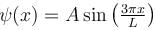 where A is a constant, what is the probability that the particle is between x = L/3 and x = 2L/3 upto 2 decimal places.
where A is a constant, what is the probability that the particle is between x = L/3 and x = 2L/3 upto 2 decimal places.
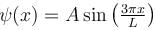 where A is a constant, what is the probability that the particle is between x = L/3 and x = 2L/3 upto 2 decimal places.
where A is a constant, what is the probability that the particle is between x = L/3 and x = 2L/3 upto 2 decimal places.Detailed Solution: Question 4
The figure shows one of the possible energy eigenfunction ψ(x) for a particle bouncing freely back and forth along the x-axis between impenetrable walls located at x ± a The potential energy equals zero for |x| < a. If the energy of the particle is 2eV when it is in the quantum state associated with its eigen function, what is its energy when it is in the quantum state of lowest energy (in eV)?
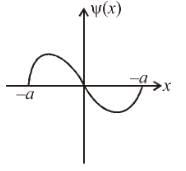

Detailed Solution: Question 5
What is the probability of finding the particle in classically forbidden region in ground state of simple harmonic oscillator
Detailed Solution: Question 6
An electron in a metal encounters a barrier layer of height 6eV and thickness 0.5 nm. If the electron energy of 5eV, what is the probability of tunneling through the barrier?
Detailed Solution: Question 7
The energy of an electron moving in one dimension in an infinitely high potential box of width 1Å.
Detailed Solution: Question 8
The solution of the schrödinger equation for the ground state of hydrogen is 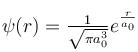 where a0 is the Bohr radius and r is the distance from the origin. The most probable value of r in units of a0.
where a0 is the Bohr radius and r is the distance from the origin. The most probable value of r in units of a0.
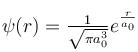 where a0 is the Bohr radius and r is the distance from the origin. The most probable value of r in units of a0.
where a0 is the Bohr radius and r is the distance from the origin. The most probable value of r in units of a0.Detailed Solution: Question 9
The density of a quantum system with two microstates is given by 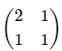 . Choose the correct option.
. Choose the correct option.
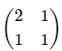 . Choose the correct option.
. Choose the correct option.Detailed Solution: Question 10


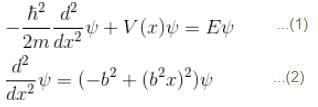
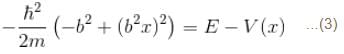
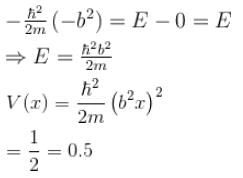
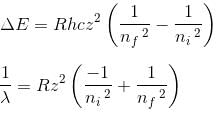


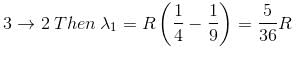
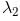 2 ---> 1
2 ---> 1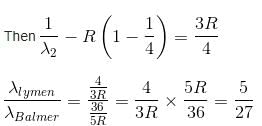

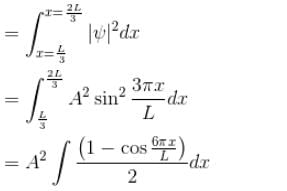
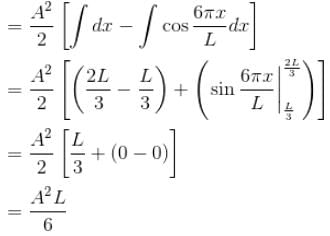
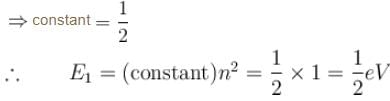
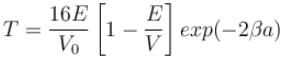
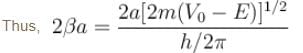
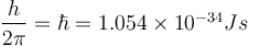
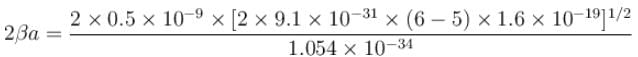
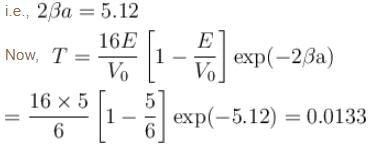
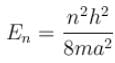
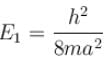 with h = 6.62 × 10–34 Js, m = 9.1 × 10–31 kg and a = 1 × 10–10 m
with h = 6.62 × 10–34 Js, m = 9.1 × 10–31 kg and a = 1 × 10–10 m