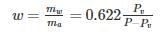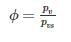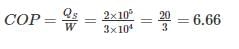Refrigeration & Air Conditioning - Mechanical Engineering MCQ
20 Questions MCQ Test - Refrigeration & Air Conditioning
In a mechanical refrigeration system, the refrigerant has the maximum temperature:
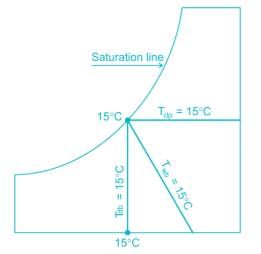
At 100% Rh, the three characteristics DBT (Dry Bulb Temperature), WBT (Wet Bulb Temperature) & DPT (Dew Point Temperature) are
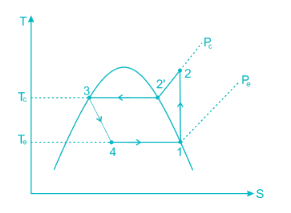
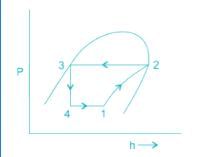
A simple saturated refrigeration cycle has the following state points. Enthalpy after compression = 425 kJ/kg; enthaly after throttling = 125 kJ/kg; enthalpy before compression = 375 kJ/kg. The COP of refrigeration is:
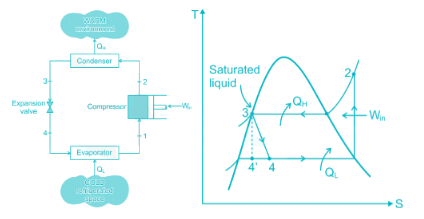
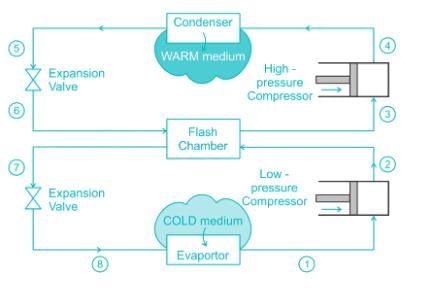
The purpose of installing a flash chamber in the refrigeration circuit is to:
If ha is enthalpy of dry air, hy is enthalpy of water vapour and w is specific humidity, the enthalpy of moist air will be
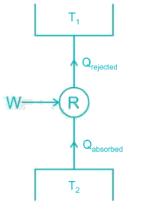
A house requires 2 × 105 kJ/h of heat for heating during winter the work done to operate heat pump is 3 × 104 kJ/h then COP will be ________
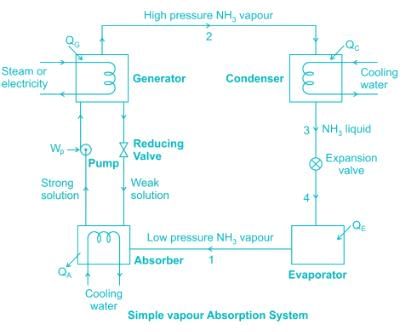
Absorption system normally uses the following refrigerant:
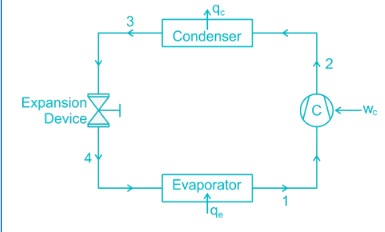
Condition of refrigerator after leaving the compressor and before entering condenser is
A Carnot cycle refrigerator operates between 250 K and 300 K. Its coefficient of performance is
The coefficient of performance is the ratio of the refrigerant effect to the _____.
A. Heat of compression
B. Work done by the compressor
C. Enthalpy increase in compressor
COP of air refrigerator is related with COP of vapour compression refrigerator as
Formation of frost on evaporator in refrigerator
1 ton of refrigeration implies heat transfer at the rate of
The throttling operation in a refrigeration cycle is carries out in:
R-12 is preferred over R-22 in deep freezer because


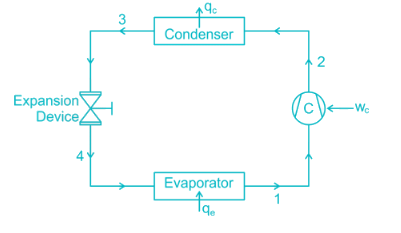
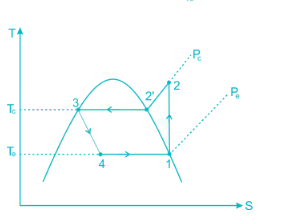
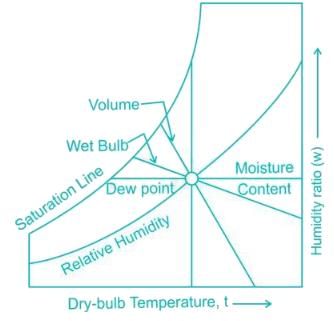

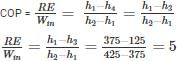
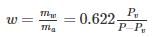
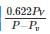 0.622PVP−Pv is used to determine
0.622PVP−Pv is used to determine