General Chemical & Magnetic Properties of d f-Block Elements - Free MCQ
MCQ Practice Test & Solutions: Test: General Chemical & Magnetic Properties of d & f-Block Elements (25 Questions)
You can prepare effectively for NEET NCERT Based Tests for NEET with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: General Chemical & Magnetic Properties of d & f-Block Elements". These 25 questions have been designed by the experts with the latest curriculum of NEET 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 35 minutes
- - Number of Questions: 25
Sign up on EduRev for free to attempt this test and track your preparation progress.
Only One Option Correct Type
Direction (Q. Nos. 1-10) This section contains 10 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONLY ONE is correct.
Q.
Of the following outer electronic configurations of atoms, the highest oxidation state is achieved by which one of them?
Detailed Solution: Question 1
Among the following series of transition metal ions, the one where all metal ions have 3d2 electronic configuration is (Atomic number, Ti = 22, V = 23, Cr = 24, Mn = 25)
[AIEEE 2004]
Detailed Solution: Question 2
For the four successive transition elements (Cr, Mn, Fe and Co), the stability of + 2 oxidation state will be there in which of the following order?
Detailed Solution: Question 3
The catalytic activity of transition metals and their compounds is mainly due to
Detailed Solution: Question 4
Many copper (I) compounds are unstable in aqueous solution and undergo disproportionation as 2Cu+ → Cu + Cu2+ . This is due to
Detailed Solution: Question 5
Gun metal is an alloy of
Detailed Solution: Question 6
In which of the following pairs both the ions are coloured in aqueous solution? (Atomic number, Sc = 21, Ti = 22, Ni = 28, Cu = 29, Co = 27)
Detailed Solution: Question 7
[Ti (H2O)6]3+ absorbs green and yellow region part of visible light. Then the transmitted colour of the compound is
Detailed Solution: Question 8
Compound of metal ion MX+(Z = 26) has a spin only magnetic moment of 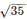 BM The number of unpaired electrons and x value in the compound are
BM The number of unpaired electrons and x value in the compound are
Detailed Solution: Question 9
In which of the following the stability of two oxidation states is correctly represented?
Detailed Solution: Question 10
One or More than One Options Correct Type
Direction (Q. Nos. 11-15) This section contains 5 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONE or MORE THAN ONE are correct.
Q.
Which one of the following arrangements represent the correct order of the property stated against it?
Detailed Solution: Question 11
The metals present in German silver alloy are
Detailed Solution: Question 12
Paramagnetic Curie temperature in Kelvin for iron is equal to :
Detailed Solution: Question 13
+8 oxidation state is/are shown by
Detailed Solution: Question 14
Ions having same colour in aqueous solution are
Detailed Solution: Question 15
Comprehension Type
Direction (Q, Nos. 16 and 17) This section contains a paragraph, describing theory, experiments, data, etc. Two questions related to the paragraph have been given. Each question has only one correct answer among the four given options (a), (b), (c) and (d).
Passage
Transition metals and their compounds have paramagnetic properties due to the presence of unpaired electrons in (n - 1)d-orbitals. The paramagnetic behaviour is expressed in terms of magnetic moment which is because of the spin of the unpaired electrons (n). It is given as
Magnetic moment = 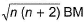
Majority of transition metal compounds are coloured both in solid state as well as in aqueous solution. This is also due to the presence of unpaired electrons in (n - 1) d-orbitals, d-orbitals splitting and d-d transition of electrons absorbing suitable visible light.
Q.
Which of the following exhibit colour due to charge transfer phenomenon, but not due to d-d transition?
Magnetic moment =
Majority of transition metal compounds are coloured both in solid state as well as in aqueous solution. This is also due to the presence of unpaired electrons in (n - 1) d-orbitals, d-orbitals splitting and d-d transition of electrons absorbing suitable visible light.
Detailed Solution: Question 16
Transition metals and their compounds have paramagnetic properties due to the presence of unpaired electrons in (n - 1)d-orbitals. The paramagnetic behaviour is expressed in terms of magnetic moment which is because of the spin of the unpaired electrons (n). It is given as
Magnetic moment = 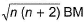
Majority of transition metal compounds are coloured both in solid state as well as in aqueous solution. This is also due to the presence of unpaired electrons in (n - 1) d-orbitals, d-orbitals splitting and d-d transition of electrons absorbing suitable visible light.
Q.
Which pair of ions are expected to be diamagnetic?
Magnetic moment =
Majority of transition metal compounds are coloured both in solid state as well as in aqueous solution. This is also due to the presence of unpaired electrons in (n - 1) d-orbitals, d-orbitals splitting and d-d transition of electrons absorbing suitable visible light.
Detailed Solution: Question 17
Match the Column I with Column II and mark the correct option from the given codes.
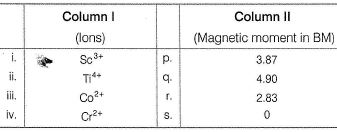
Detailed Solution: Question 18
Match the Column I with Column II and mark the correct option from the given codes.
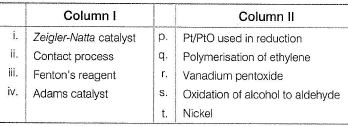
Detailed Solution: Question 19
One Integer Value Correct Type
Direction (Q. Nos. 21-24) This section contains 4 questions. When worked out will result in an integer from 0 to 9 (both inclusive).
Q.
Number of alloys that contain nickel among the following solder, gun metal, German silver, nichrome, monel metal, constanton, bell metal, duralumin, type metal, invar, alnico.
Detailed Solution: Question 20
Green vitriol is FeSO4.xH2O and white vitriol is ZnSO4.yH2O, Then, the values of x and y are
Detailed Solution: Question 21
Oxidation state of chromium in CrO5 is +x. Here, value of x is
Detailed Solution: Question 22
Number of compounds in which metal has zero oxidation state
WO3,Ni(CO)4, MoO3, Fe(CO)5, Cr(CO)6, [Pt(NH3)2CI2], Co2(CO)8 and Mn2(CO)10
Detailed Solution: Question 23
Statement Type
Direction (Q. No. 25) This section is based on Statement I and Statement II. Select the correct answer from the codes given below.
Q.
Statement l :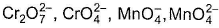 ,Cu2O and Ni-DMG complex are coloured.
,Cu2O and Ni-DMG complex are coloured.
Statement II : It is due to charge transfer transition.
Detailed Solution: Question 24
The oxidation state of Ni in [Ni(CO)₄] is:
Detailed Solution: Question 25


