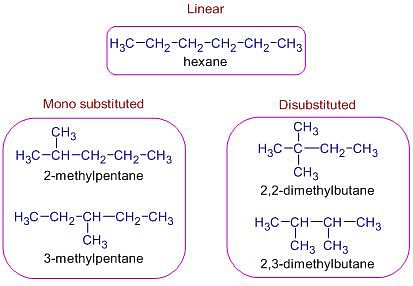
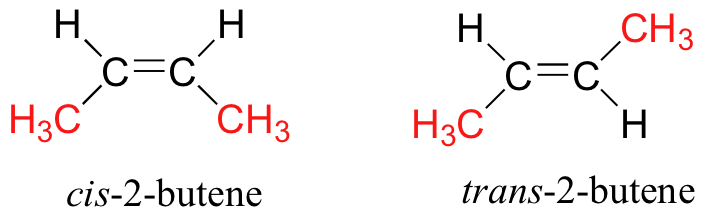
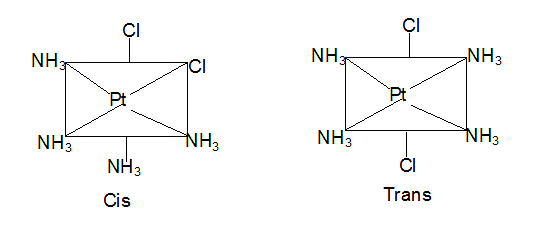
NEET Exam > NEET Tests > Chemistry Class 11 > Test: Isomerism - NEET MCQ
Test: Isomerism - NEET MCQ
Test Description
10 Questions MCQ Test Chemistry Class 11 - Test: Isomerism
Test: Isomerism for NEET 2025 is part of Chemistry Class 11 preparation. The Test: Isomerism questions and answers have been
prepared according to the NEET exam syllabus.The Test: Isomerism MCQs are made for NEET 2025 Exam. Find important
definitions, questions, notes, meanings, examples, exercises, MCQs and online tests for Test: Isomerism below.
Solutions of Test: Isomerism questions in English are available as part of our Chemistry Class 11 for NEET & Test: Isomerism solutions in
Hindi for Chemistry Class 11 course. Download more important topics, notes, lectures and mock
test series for NEET Exam by signing up for free. Attempt Test: Isomerism | 10 questions in 15 minutes | Mock test for NEET preparation | Free important questions MCQ to study Chemistry Class 11 for NEET Exam | Download free PDF with solutions
Detailed Solution for Test: Isomerism - Question 1
Test: Isomerism - Question 2
2-chloropropane and 1-chloropropane exhibit ____________ isomerism.
Detailed Solution for Test: Isomerism - Question 2
Detailed Solution for Test: Isomerism - Question 3
Detailed Solution for Test: Isomerism - Question 4
Detailed Solution for Test: Isomerism - Question 5
Detailed Solution for Test: Isomerism - Question 6
Detailed Solution for Test: Isomerism - Question 7
Detailed Solution for Test: Isomerism - Question 8
Test: Isomerism - Question 9
What is the specific rotation if its observed rotation is given as 3x, its length is given as x and density is given as 3/y?
Detailed Solution for Test: Isomerism - Question 9
Detailed Solution for Test: Isomerism - Question 10
|
127 videos|245 docs|87 tests
|
Information about Test: Isomerism Page
In this test you can find the Exam questions for Test: Isomerism solved & explained in the simplest way possible.
Besides giving Questions and answers for Test: Isomerism, EduRev gives you an ample number of Online tests for practice