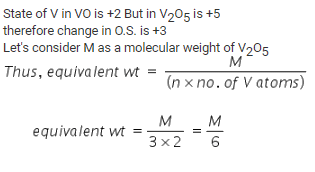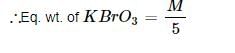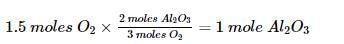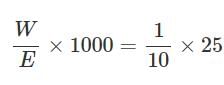Mole Concept, Volumetric & Redox - 1 - Free MCQ Practice Test with solutions,
MCQ Practice Test & Solutions: Test: Mole Concept, Volumetric & Redox - 1 (20 Questions)
You can prepare effectively for Chemistry Physical Chemistry with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Mole Concept, Volumetric & Redox - 1". These 20 questions have been designed by the experts with the latest curriculum of Chemistry 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 90 minutes
- - Number of Questions: 20
Sign up on EduRev for free to attempt this test and track your preparation progress.
4.4g of CO2 and 2.24 litre of H2 at STP (273.15 K and 1 atm pressure) are mixed in a container. The total of molecules present in the container will be
Detailed Solution: Question 1
4I– + Hg2+ → HgI42– ; 1 mole of each Hg2+ and I– will form:
Detailed Solution: Question 2
How many grams of I2 are present in a solution which requires 40 ml of 0.11N Na2S2O3 to react with it via the reaction:
S2O32– + I2 → S4O62– + 2I–?
S2O32– + I2 → S4O62– + 2I–?
Detailed Solution: Question 3
Two elements A (atomic mass = 75) and B (atomic mass = 16) combine to yield a compound. The % by weight of A in the compound was found to be 75.08. The formula of compound is:
Detailed Solution: Question 4
What weight of HNO3 is needed to convert 5g of Iodine into Iodic acid according to the reaction:
I2 + HNO3→ HIO3 + NO2 +H2O
I2 + HNO3→ HIO3 + NO2 +H2O
Detailed Solution: Question 5
In the reaction, VO + Fe2O3 → FeO + V2O5, the equilvalent weight of V2O5 is equal to its
Detailed Solution: Question 6
When BrO3– ion reacts with Br– ion in acid solution Br2 is liberated. The eq. weight of KBrO3 in this reaction is:
10e + 2Br5+ → Br02
Detailed Solution: Question 7
The hydrated salt, Na2SO4.nH2O, undergoes 55.9 % loss in weight on heating and become anhydrous. The value of n will be:
Detailed Solution: Question 8
When a metal is burnt, its weight is increased by 24%. The eq. weight of the metal will be:
Detailed Solution: Question 9
One mole of potassium chlorate is thermally decomposed and excess of aluminum is burnt in the gaseous product .How many moles of aluminum oxide are formed?
Detailed Solution: Question 10
How much Cl2 at STP is liberated when one mole of KMnO4 reacts with HCl ?
Detailed Solution: Question 11
Minimum quantity of H2S needed to precipitate 63.5g of Cu2+ is nearly:
Detailed Solution: Question 12
2g of CaCO3 was treated with 0.1M HCl (500 ml). The volume of CO2 evolved at STP after heating the solution is:
10g of limestone on heating produces 4.2g of CaO. the percentage purity of CaCO3 in limestone is:
Detailed Solution: Question 14
‘x’ g of KClO3 on decomposition gives ‘y’ ml of O2 at STP. The % purity of KClO3 would be
Detailed Solution: Question 15
33.6g of an impure sample of sodium bicarbonate when heated strongly gave 4.4g of CO2. The % purity of NaHCO3 would be:
Detailed Solution: Question 16
0.16g of dibasic acid required 25ml of N/10 NaOH for complete neutralization. Molecular wt. of acid is:
Detailed Solution: Question 17
The molar coefficients of AsO33– and MnO4- in the reaction are:
AsO33– + MnO4Θ → AsO43– + MnO2
Detailed Solution: Question 18
Which of the following is a disproportionation reaction?
Detailed Solution: Question 19
Hydrogen and oxygen combine to form H2O2 and H2O containing 5.93% and 11.2 % hydrogen respectively. The data illustrates:
Detailed Solution: Question 20
84 videos|147 docs|67 tests |


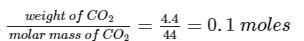
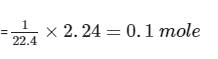
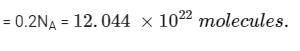

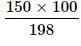 = 75.08g of A
= 75.08g of A