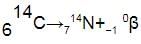Nuclei: Radioactivity - Free MCQ Practice Test with solutions, NEET Physics
MCQ Practice Test & Solutions: Test: Nuclei: Radioactivity (5 Questions)
You can prepare effectively for NEET Physics Class 12 with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Nuclei: Radioactivity". These 5 questions have been designed by the experts with the latest curriculum of NEET 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 10 minutes
- - Number of Questions: 5
Sign up on EduRev for free to attempt this test and track your preparation progress.
Detailed Solution: Question 1
A sample of radioactive material contains 1018 atoms. The half life of the material is 2 days, then the activity of the sample is
Detailed Solution: Question 2
Detailed Solution: Question 3
When a radioactive nucleus emits a β – particle, the mass number of the atom
Detailed Solution: Question 4
The number of protons or atomic number is reduced to 2 by which form of radioactive decay?
Detailed Solution: Question 5
74 videos|347 docs|82 tests |