Oxidation Reduction & Disproportionation Reactions - Free MCQ Practice
MCQ Practice Test & Solutions: Test: Oxidation Reduction & Disproportionation Reactions (23 Questions)
You can prepare effectively for NEET Chemistry Class 11 with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Oxidation Reduction & Disproportionation Reactions". These 23 questions have been designed by the experts with the latest curriculum of NEET 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 35 minutes
- - Number of Questions: 23
Sign up on EduRev for free to attempt this test and track your preparation progress.
Direction (Q. Nos. 1-18) This section contains 18 multiple choice questions. Each question has four
choices (a), (b), (c) and (d), out of which ONLY ONE option is correct.
Q. Oxidation can be defined as the terms
I. gain of electron and hydrogen
II. gain of oxygen and loss of electron
III. increase in oxidation number
IV. decrease in oxidation number
Select the correct terms
choices (a), (b), (c) and (d), out of which ONLY ONE option is correct.
II. gain of oxygen and loss of electron
III. increase in oxidation number
IV. decrease in oxidation number
Detailed Solution: Question 1
Reduction is defined in terms of
I. electronation and hydrogenation
II. deelectronation and gain of oxygen
III. increase in oxidation number
IV. decrease in oxidation number
Select the correct terms
II. deelectronation and gain of oxygen
III. increase in oxidation number
IV. decrease in oxidation number
Detailed Solution: Question 2
In balancing the half-reaction, CN- → CNO-
The number of electrons that must be added is
Detailed Solution: Question 3
Select the set of compounds with oxidation-reduction duality.
Detailed Solution: Question 4
In the following balanced reaction,
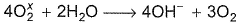
Detailed Solution: Question 5
I− reduces IO3- and I2 and itself oxidised to I2 in acidic medium. Thus, final reaction is
Detailed Solution: Question 6
Detailed Solution: Question 7
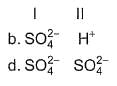
Consider the following reactions,
I. Zn + dil. H2SO4 → ZnSO4 + H2
II. Zn + conc. H2SO4 → ZnSO4+ SO2 + H2O
Oxidising agents in I and II are
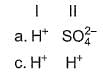
II. Zn + conc. H2SO4 → ZnSO4+ SO2 + H2O
Oxidising agents in I and II are
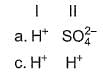

Detailed Solution: Question 8
In the following reactions except in one, oxygen is the reducing agent. Exceptional reaction is
Detailed Solution: Question 9
In the following reaction,
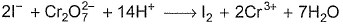
Which ions are not balanced?
Detailed Solution: Question 10
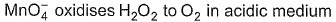
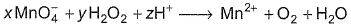
Coefficient x, y and z are respectively

Detailed Solution: Question 11
Which of the following species does not show disproportionation reaction?
Detailed Solution: Question 12
Which is the intramolecular oxidation-reduction reaction?
Detailed Solution: Question 13
Consider the following experimental facts,
I. When Cl2 gas is passed into Kl solution containing CHCI3, violet colour appears in CHCI3 layer.
II. When Cl2 gas is passed into KBr solution containing CHCI3, orange colour appears in CHCI3 layer.
III. When Cl2 gas is passed into a solution containing KBr, Kl and KCI, containing CHCI3, violet colour appears in CHCI3 layer.
Select the correct experimental facts.
II. When Cl2 gas is passed into KBr solution containing CHCI3, orange colour appears in CHCI3 layer.
III. When Cl2 gas is passed into a solution containing KBr, Kl and KCI, containing CHCI3, violet colour appears in CHCI3 layer.
Detailed Solution: Question 14
Based on the following reaction,
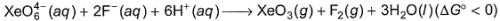
It can be concluded that
Detailed Solution: Question 15
For the redox reaction,
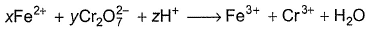
x, y and z are
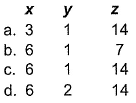
x, y and z are
Detailed Solution: Question 16
In the following conversion of chromite into soluble chromium salt
4Fe(CrO2)2+ 8Na2CO3 + 7O2 --> 2Fe2O3 + 8Na2CrO4 + 8CO2
There is
4Fe(CrO2)2+ 8Na2CO3 + 7O2 --> 2Fe2O3 + 8Na2CrO4 + 8CO2
Detailed Solution: Question 17
In which of the following reactions H2O2 acts as a reducing agent?
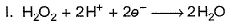
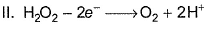
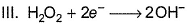
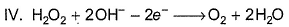
Detailed Solution: Question 18
Consider the following reaction,
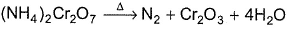
Detailed Solution: Question 19
Following reaction, 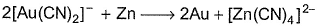 is an example of
is an example of
Detailed Solution: Question 20
Select the correct statement(s).
Detailed Solution: Question 21
For the reaction, 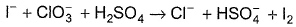
The correct statement(s) in the balanced equation is/are
[JEE Advanced 2014]
Detailed Solution: Question 22
Identify intramolecular oxidation-reduction reaction.
Detailed Solution: Question 23
97 videos|243 docs|71 tests |



