Properties of D-block elements - Free MCQ Practice Test with solutions,
MCQ Practice Test & Solutions: Test: Properties of D-block elements (20 Questions)
You can prepare effectively for JEE Chemistry for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Properties of D-block elements". These 20 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 20 minutes
- - Number of Questions: 20
Sign up on EduRev for free to attempt this test and track your preparation progress.
Which of the following is not a condition for complex formation?
Detailed Solution: Question 1
Which one of the following is a diamagnetic ion?
Detailed Solution: Question 2
The inner transition elements are the elements in which the added electrons go to:
Detailed Solution: Question 3
Which of the following pairs of ions are colourless?
Detailed Solution: Question 4
Which of the following is not correct about transition metals?
Detailed Solution: Question 5
Zinc and mercury do not show variable valency like 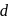 -block element because
-block element because
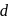 -block element because
-block element becauseDetailed Solution: Question 6
Which one of the following is diamagnetic ion?
Detailed Solution: Question 7
Which group contains coloured ions out of
1. Cu2+
2. Ti4+
3. Co2+
4. Fe2+
1. Cu2+
2. Ti4+
3. Co2+
4. Fe2+
Detailed Solution: Question 8
A compound of a metal ion 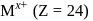 has a spin only magnetic moment of
has a spin only magnetic moment of 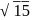 Bohr Magnetons. The number of unpaired electrons in the compound are
Bohr Magnetons. The number of unpaired electrons in the compound are
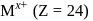 has a spin only magnetic moment of
has a spin only magnetic moment of 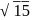 Bohr Magnetons. The number of unpaired electrons in the compound are
Bohr Magnetons. The number of unpaired electrons in the compound areDetailed Solution: Question 9
Cuprous ion is colourless while cupric ion is coloured because
Detailed Solution: Question 10
Among the following, the compound that is both paramagnetic and coloured, is
Detailed Solution: Question 11
Mercury is the only metal which is liquid at 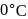 . This is due to its
. This is due to its
Detailed Solution: Question 12
Which of the following pair of ions have same paramagnetic moment?
Detailed Solution: Question 13
The correct order of decreasing second ionisation enthalpy of Ti (22), V (23), Cr (24) and Mn (25) is-
Detailed Solution: Question 14
Which pair of ions have same magnetic moment?
Detailed Solution: Question 15
The correct statement about Cr2+ and Mn3+ among the following is (Given, atomic numbers of Cr = 24 and Mn = 25)
Detailed Solution: Question 16
A transition element containing only one electron in 4s-orbital is:
Detailed Solution: Question 17
Which of the following characteristics of the transition metals is associated with their catalytic activity?
Detailed Solution: Question 18
How many unpaired electrons will be present in the ground state of an atom which has valence electronic configuration 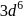 in its +3 oxidation state?
in its +3 oxidation state?
Detailed Solution: Question 19
Which of the following is the common oxidation state for 'transition' elements?
Detailed Solution: Question 20
335 videos|699 docs|300 tests |



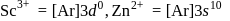
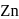 and
and 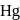 have
have 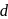 -shells completely filled hence they do not exhibit variable valency.
-shells completely filled hence they do not exhibit variable valency.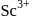 is diamagnetic as it does not contain any unpaired electron while others contain.
is diamagnetic as it does not contain any unpaired electron while others contain.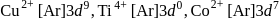
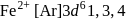 are coloured ions
are coloured ions .
.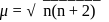 where
where 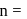 number of unpaired electrons
number of unpaired electrons 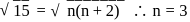
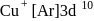 there is no unpaired electron,
there is no unpaired electron, 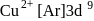 contains one unpaired electron hence coloured.
contains one unpaired electron hence coloured.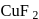 is both paramagnetic and coloured.
is both paramagnetic and coloured.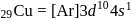
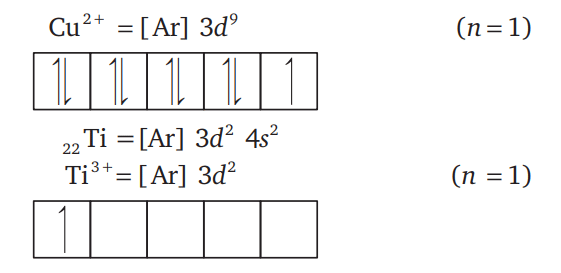
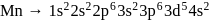
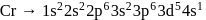
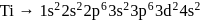
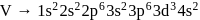
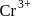 magnetic moment as 3.87
magnetic moment as 3.87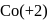 magnetic moment as 3.87
magnetic moment as 3.87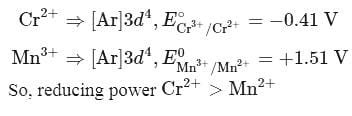
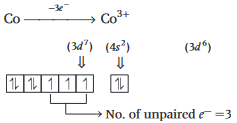
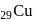 has the electronic structure of
has the electronic structure of 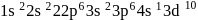 . One electron moves from the 45 -orbital to attain a more stable state with completely filled
. One electron moves from the 45 -orbital to attain a more stable state with completely filled 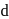 orbital.
orbital.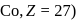 will be
will be 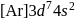 .
.
 -orbital, and removing the electrons from the sub shell below it requires immense amounts of energy, this limits them to a +2 charge most of the time. Hence, the correct option is (c).
-orbital, and removing the electrons from the sub shell below it requires immense amounts of energy, this limits them to a +2 charge most of the time. Hence, the correct option is (c).