SN2 Reaction Basics - Free MCQ Practice Test with solutions, NEET Chemistry
MCQ Practice Test & Solutions: Test: SN2 Reaction Basics (20 Questions)
You can prepare effectively for NEET Chemistry Class 12 with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: SN2 Reaction Basics". These 20 questions have been designed by the experts with the latest curriculum of NEET 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 30 minutes
- - Number of Questions: 20
Sign up on EduRev for free to attempt this test and track your preparation progress.
Only One Option Correct Type
Direction (Q. Nos. 1-8) This section contains 8 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONLY ONE is correct.
Q.
Which of the SN2 reaction is fastest?
Detailed Solution: Question 1
Detailed Solution: Question 2
A correct statement about transition state of SN2 reaction is
Detailed Solution: Question 3
What is the correct increasing order of reactivity of the followings in SN2 reaction ?
I. CH2 = CHCH2 — Br
II. CH2 = CH— I
III. CH3CH2CH2 — I
IV. CH3OCH2CH2 — I
II. CH2 = CH— I
III. CH3CH2CH2 — I
IV. CH3OCH2CH2 — I
Detailed Solution: Question 4
What is the correct increasing order of reactivity of the following in the SN2 reaction?

Detailed Solution: Question 5
Consider the two lines shown in the diagram given below.
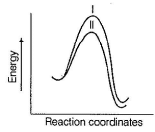
Q.
In a SN2 reaction, these two lines compare the effect of the
Detailed Solution: Question 6
Consider the two lines shown in the diagram given below.

Q.
Which of the following apply appropriately to a SN2 reaction?
Detailed Solution: Question 7
Detailed Solution: Question 8
One or More than One Options Correct Type
Direction (Q. Nos. 9-14) This section contains 6 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONE or MORE THAN ONE are correct.
Q.
In a SN2 reaction rate of reaction depends on
Detailed Solution: Question 9
Consider the following SN2 reaction,
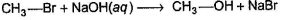
Q.
Which of the following could increase the reactivity (rate) of reaction ?
Detailed Solution: Question 10
A SN2 reaction involves back side attack of nucleophile at the α-carbon of substrate because
Detailed Solution: Question 11
1-chlorobutane is more reactive than 2-chloro-2-methyl propane in a SN2 reaction because
Detailed Solution: Question 12
Consider the reaction given below.
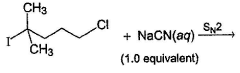
Q.
The correct statement(s) applicable to the above reaction is/are
Detailed Solution: Question 13
Detailed Solution: Question 14
Comprehension Type
Direction (Q. Nos. 15-17) This section contains a paragraph, describing theory, experiments, data, etc. Three Questions related to the paragraph have been given. Each question has only one correct answer among the four given options (a), (b), (c) and (d).
The general mechanism of a SN2 reaction is as follows.
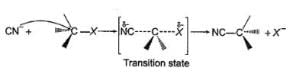
Factors that limit the rate of reaction are steric hindrance at α-cabron of substrate and strength of C—X bond. Any factor which stabilises transition state increases the rate of reaction.
Q. Consider the following SN2 reaction,
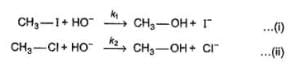
Which of the following energy diagram is correctly labelled?


Detailed Solution: Question 15
The general mechanism of a SN2 reaction is as follows.
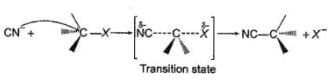
Factors that limit the rate of reaction are steric hindrance at α-cabron of substrate and strength of C—X bond. Any factor which stabilises transition state increases the rate of reaction.
Q.
In the previous question, under identical reaction condition, i.e. tem perature, concentration of substrate and nucleophile, the correct relationship between rate constant is

Detailed Solution: Question 16
The general mechanism of a SN2 reaction is as follows.
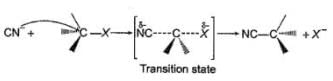
Factors that limit the rate of reaction are steric hindrance at α-cabron of substrate and strength of C—X bond. Any factor which stabilises transition state increases the rate of reaction.
Q.
Which of the following is not a correct representation of SN2 reaction ?

Detailed Solution: Question 17
One Integer Value Correct Type
Direction (Q. Nos. 18-20) This section contains 3 questions. When worked out will result in an integer from 0 to 9 (both inclusive).
Consider the following reaction,
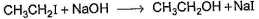
Q.
The above reaction was started taking equal concentrations of ethyl iodide and NaOH. After 1.0 h, concentration of iodoethane is dropped to (1/3)rd of initial value. By what factor, the rate of reaction would have been decreased by the same time?
Detailed Solution: Question 18
Under identical experimental condition, how many of the following substrate react at faster rate with aqueous NaOH than 2-bromobutane as substrate?










Detailed Solution: Question 19
Consider the following compound,
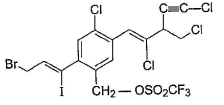
Q. If the above compound is treated with excess of NaCN(aq), how many CN- group would be incorporated by SN2 reaction?
Detailed Solution: Question 20
54 videos|290 docs|74 tests |


