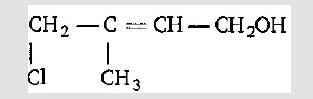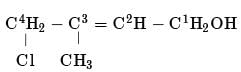Carbon and its compounds (Easy) - Free MCQ Practice Test with solutions,
MCQ Practice Test & Solutions: Test: Carbon and its compounds (Easy) (15 Questions)
You can prepare effectively for Class 10 with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Carbon and its compounds (Easy)". These 15 questions have been designed by the experts with the latest curriculum of Class 10 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 15 minutes
- - Number of Questions: 15
Sign up on EduRev for free to attempt this test and track your preparation progress.
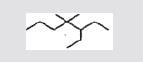
Alcohols may be represented by the general formula:
Detailed Solution: Question 1
Detailed Solution: Question 2
Detailed Solution: Question 3
Detailed Solution: Question 4
Which of the following structures correctly represents the electron dot structure of a chlorine molecule?
Detailed Solution: Question 5
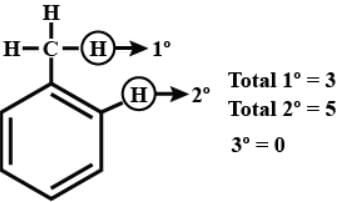
How many 1°, 2° and 3° H atoms are present in (Toluene ) respectively.
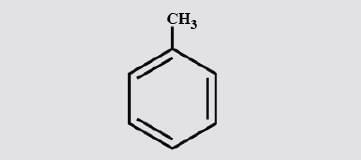

Detailed Solution: Question 6
For Carbocation the Stability Order Will be _________________.
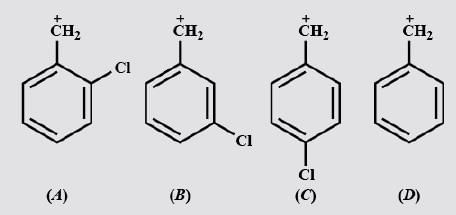

Detailed Solution: Question 7
Graphite differs from diamond because graphite is:
This question has multiple correct options
Detailed Solution: Question 8
The IUPAC name of the compound CH3CH = CHC = CH is:
Detailed Solution: Question 9
Detailed Solution: Question 10
The process of making soap by the hydrolysis of fats and oils with alkalis is called:
Detailed Solution: Question 11
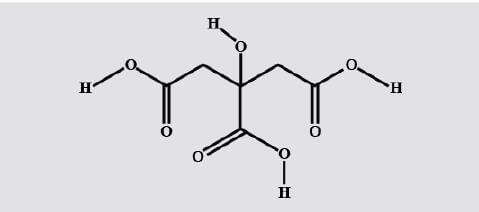
Trivial name of 2, 3-dihydroxy butanedioic acid is-
Detailed Solution: Question 12
Detailed Solution: Question 13
Which of the following is not a unit of energy
Detailed Solution: Question 14
Detailed Solution: Question 15