Atoms and Molecules- Case Based Type Questions - Free with solutions
MCQ Practice Test & Solutions: Test: Atoms and Molecules- Case Based Type Questions (15 Questions)
You can prepare effectively for Class 9 Class 9: Additional Practice with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Atoms and Molecules- Case Based Type Questions". These 15 questions have been designed by the experts with the latest curriculum of Class 9 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 30 minutes
- - Number of Questions: 15
Sign up on EduRev for free to attempt this test and track your preparation progress.
Direction: The following data represents the distribution of electrons, protons and neutrons in atoms of four elements A, B, C, D. Understand the data carefully and answer the following questions.

Q. Select the correct electronic distribution of element B:

Detailed Solution: Question 1
Direction: The following data represents the distribution of electrons, protons and neutrons in atoms of four elements A, B, C, D. Understand the data carefully and answer the following questions.
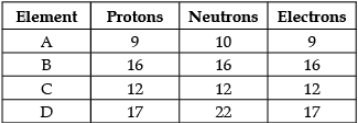
Q. The Mass number of element D is:

Detailed Solution: Question 2
Direction: The following data represents the distribution of electrons, protons and neutrons in atoms of four elements A, B, C, D. Understand the data carefully and answer the following questions.
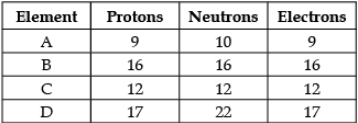
Q. The atomic number of element B is:

Detailed Solution: Question 3
Direction: The following data represents the distribution of electrons, protons and neutrons in atoms of four elements A, B, C, D. Understand the data carefully and answer the following questions.
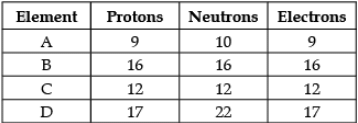
Q. The Valency of element A:

Detailed Solution: Question 4
Direction: Sanjana observed that when 3.0 gm of carbon is burnt in 8.0 gm of oxygen, 11.0 gm of carbon dioxide is produced. Based on the given information, answer the following questions.
Q. Name the Law of Chemical Combination shown in the above passage:
Detailed Solution: Question 5
Direction: Sanjana observed that when 3.0 gm of carbon is burnt in 8.0 gm of oxygen, 11.0 gm of carbon dioxide is produced. Based on the given information, answer the following questions.
Q. In what ratio does carbon and oxygen combine to form carbon dioxide?
Detailed Solution: Question 6
Direction: Sanjana observed that when 3.0 gm of carbon is burnt in 8.0 gm of oxygen, 11.0 gm of carbon dioxide is produced. Based on the given information, answer the following questions.
Q. In a compound water at what ratio hydrogen and oxygen combine to form water:
Detailed Solution: Question 7
Direction: Sanjana observed that when 3.0 gm of carbon is burnt in 8.0 gm of oxygen, 11.0 gm of carbon dioxide is produced. Based on the given information, answer the following questions.
Q. In a chemical substance, elements are present in a definite proportion by ______.
Detailed Solution: Question 8
Direction: Two class students of class 9th, Aashi and Sheena, were asked to take 5.3 g of sodium carbonate and 6 g of ethanoic acid to make 2.2 g of carbon dioxide, 0.9 g of water and 8.2 g of sodium ethanoate. Aashi followed the instructions but Sheena took the chemicals without measuring their amounts.
Q. Which law does this agreements shows?
Detailed Solution: Question 9
Direction: Two class students of class 9th, Aashi and Sheena, were asked to take 5.3 g of sodium carbonate and 6 g of ethanoic acid to make 2.2 g of carbon dioxide, 0.9 g of water and 8.2 g of sodium ethanoate. Aashi followed the instructions but Sheena took the chemicals without measuring their amounts.
Q. Whose activity do you think will be in agreement with the law?
Detailed Solution: Question 10
Direction: Two class students of class 9th, Aashi and Sheena, were asked to take 5.3 g of sodium carbonate and 6 g of ethanoic acid to make 2.2 g of carbon dioxide, 0.9 g of water and 8.2 g of sodium ethanoate. Aashi followed the instructions but Sheena took the chemicals without measuring their amounts.
Q. The Law states that _______ can neither be created nor be destroyed in a chemical reaction.
Detailed Solution: Question 11
Direction: Rahul took 5 moles of carbon atoms in a container and Sohan also took 5 moles of sodium atoms in another container of same weight.
Q. Whose container has more number of atoms?
Detailed Solution: Question 12
Direction: Rahul took 5 moles of carbon atoms in a container and Sohan also took 5 moles of sodium atoms in another container of same weight.
Q. Which container is heavier?
Detailed Solution: Question 13
Direction: Rahul took 5 moles of carbon atoms in a container and Sohan also took 5 moles of sodium atoms in another container of same weight.
Q. Number of atoms in one mole:
Detailed Solution: Question 14
Direction: Rahul took 5 moles of carbon atoms in a container and Sohan also took 5 moles of sodium atoms in another container of same weight.
Q. The exact number of atoms present in 12 gm of Carbon-12:
Detailed Solution: Question 15
4 docs|108 tests |


