Important Questions (1 mark): Structure of the Atom - Free MCQ Test
MCQ Practice Test & Solutions: Important Questions (1 mark): Structure of the Atom (25 Questions)
You can prepare effectively for Class 9 Online MCQ Tests for Class 9 with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Important Questions (1 mark): Structure of the Atom". These 25 questions have been designed by the experts with the latest curriculum of Class 9 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 25 minutes
- - Number of Questions: 25
Sign up on EduRev for free to attempt this test and track your preparation progress.
Which of the following is regarded as a universal particle?
Detailed Solution: Question 1
The main subatomic particles of nucleus are-
Detailed Solution: Question 2
What is name given to the number of protons in the nucleus of the atom?
Detailed Solution: Question 3
The atom which does not contain any neutron in the nucleus is
Detailed Solution: Question 4
Detailed Solution: Question 5
Match the following with correct response.


There are 14 protons and 13 neutrons on the nucleus of an element. What is its mass number?
Detailed Solution: Question 7
“Electrons present in the extra nuclear portion are not stationary rather they revolve around the nucleus at high speed following a circular path” Who said this?
Detailed Solution: Question 8
Match the following with correct response.


Nitrogen atom has atomic mass of 14 u and has 7 protons in its nucleus. How many neutrons does it have?
Detailed Solution: Question 10
Match the following with correct response.


In an alpha scattering experiment, few alpha particles rebounded because
Detailed Solution: Question 12
Match the following with correct response.
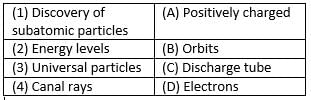

Detailed Solution: Question 14
Isotopes of an element have
A. Same chemical properties
B. Same atomic masses
C. Differeat atomic numbers
D. Atoms of same element
A. Same chemical properties
B. Same atomic masses
C. Differeat atomic numbers
D. Atoms of same element
Detailed Solution: Question 15
How many times is radius of extra nuclear portion more than that of the nucleus of an atom?
Detailed Solution: Question 16
Match the following with correct response.
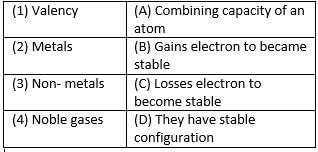

An atom has a mass number of 23 and atomic number 11. The number of protons are_________.
Detailed Solution: Question 18
How many electrons are present in the species He2+ion?
Detailed Solution: Question 19
Who got the Noble Prize for the work on the structure of atom?
Detailed Solution: Question 20
Which of the following is not observed by Rutherford in the α-particle scattering experiment?
A. Most of the α-particle rebound after hitting the gold foil
B. Some of the particles deflect by their path
C. Some of the particles did not pass through the gold foil
D. Most of the particles pass straight through the gold foil
A. Most of the α-particle rebound after hitting the gold foil
B. Some of the particles deflect by their path
C. Some of the particles did not pass through the gold foil
D. Most of the particles pass straight through the gold foil
Detailed Solution: Question 21
Detailed Solution: Question 22
In a species, the number of electrons is more than the number of protons. Predict its nature.
Detailed Solution: Question 23
Out of L and M shells which is near to the nucleus of an atom?
Detailed Solution: Question 24
Which of the following is correct about atom?
A. It is the smallest unit of matter
B. Size of atom is very large as compared to the nucleus of the atom
C. In an atom number of protons is equal to number of electrons
D. Atoms are stable
A. It is the smallest unit of matter
B. Size of atom is very large as compared to the nucleus of the atom
C. In an atom number of protons is equal to number of electrons
D. Atoms are stable
Detailed Solution: Question 25
5 docs|319 tests |


