VITEEE Chemistry Test - 6 - JEE MCQ
30 Questions MCQ Test VITEEE: Subject Wise and Full Length MOCK Tests - VITEEE Chemistry Test - 6
Which of the following process is employed to convert alkyl halide into alcohol?
Which of the following compounds will undergo self aldol condensation in the presence of cold dilute alkali?
| 1 Crore+ students have signed up on EduRev. Have you? Download the App |
An organic compound 'A' has the molecular formula C₃H₆O. It undergoes iodoform test. When saturated with HCl it gives 'B' of molecular formula C₉H₁₄O. 'A' and 'B' respectively are
Irrespective of the source, pure sample of water always yields 88.89% mass of oxygen and 11.11% mass of hydrogen. This is explained by the law of
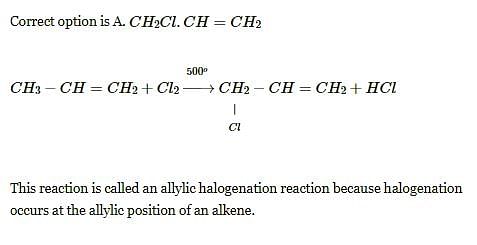
How many monochlorobutanes will be obtained on chlorination of n-butane?
Which one of the following esters is obtained by the esterification of propan-2-ol with ethanoic acid?
A is a higher phenol and B is an aromatic carboxylic acid. Separation of a mixture of A and B can be carried out easily by having a solution
A chemical reaction is catalyzed by a catalyst X. Hence X
A glass bulb is filled with NO₂ gas and immersed in an ice bath at 0oC which becomes colourless after some time . This colourless gas will be
The Δ H value for the reaction H2 + Cl2 → 2HCl is − 44.12 K.cal. If E1 is the activation energy of the reactants and E2 is the activation energy of the products, then for the above reaction
Calculate the heat of formation Δ H of CO (in kcal) from the following data :
C(graphite) + O2 (g) → CO2 (g) ; Δ H = − 94 K . c a l
CO(g) + 1/2O2 (g) → CO2 (g) ; Δ H = − 68 K . c a l
The pair in which both species have the same magnetic moment (spin only value) is :
When benzenediazonium chloride in hydrochloric acid reacts with cuprous chloride, then chlorobenzene is formed. The reaction is called
96500 C electricity is passed through CuSO₄. The amount of copper precipitated is
The element, which forms oxides in all oxidation states + 1 to + 5, is
Acetic anhydride reacts with diethyl ether in the presence of anhydrous AlCl₃ to give
Which of the following metal is leached by cyanide process?
At which one of the following temperature-pressure conditions, the deviation of a gas from ideal behaviour is expected to be minimum ?
|
1 videos|2 docs|73 tests
|
|
1 videos|2 docs|73 tests
|